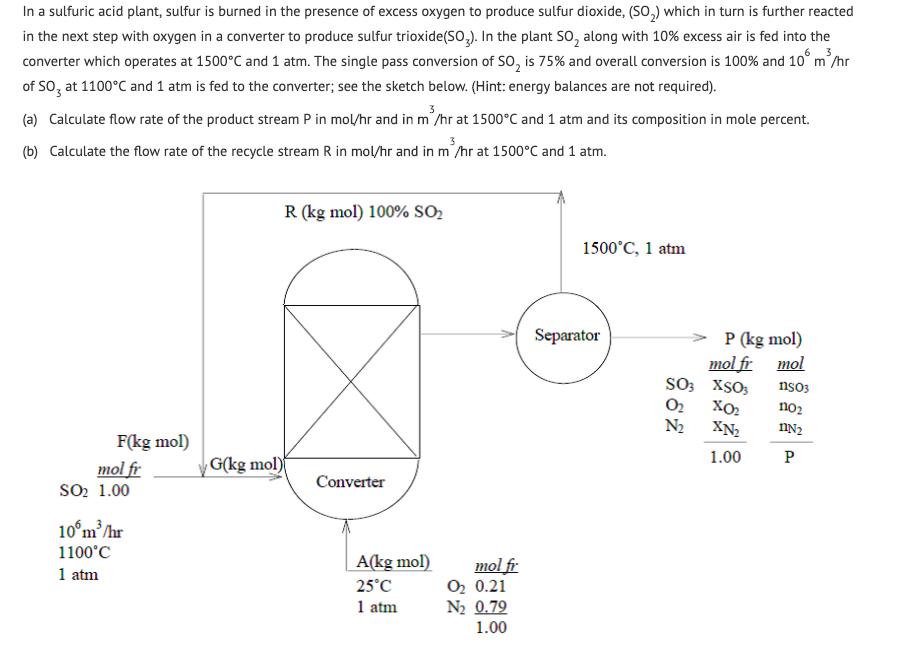
In a sulfuric acid plant, sulfur is burned in the presence of excess oxygen to produce...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
In a sulfuric acid plant, sulfur is burned in the presence of excess oxygen to produce sulfur dioxide, (SO,) which in turn is further reacted in the next step with oxygen in a converter to produce sulfur trioxide(SO,). In the plant SO, along with 10% excess air is fed into the converter which operates at 1500°C and 1 atm. The single pass conversion of SO, is 75% and overall conversion is 100% and 10° m'/hr of SO, at 1100°C and 1 atm is fed to the converter; see the sketch below. (Hint: energy balances are not required). (a) Calculate flow rate of the product stream P in mol/hr and in m'/hr at 1500°C and 1 atm and its composition in mole percent. (b) Calculate the flow rate of the recycle stream R in mol/hr and in m'/hr at 1500°C and 1 atm. R (kg mol) 100% SO2 1500°C, 1 atm Separator P (kg mol) mol fr mol SO3 XSO3 O2 N2 nso3 no2 XN2 F(kg mol) 1.00 P G(kg mol) mol fr Soo 1.00 Converter 10°m/hr 1100°C A(kg mol) mol fr O2 0.21 N2 0.79 1.00 1 atm 25°C 1 atm In a sulfuric acid plant, sulfur is burned in the presence of excess oxygen to produce sulfur dioxide, (SO,) which in turn is further reacted in the next step with oxygen in a converter to produce sulfur trioxide(SO,). In the plant SO, along with 10% excess air is fed into the converter which operates at 1500°C and 1 atm. The single pass conversion of SO, is 75% and overall conversion is 100% and 10° m'/hr of SO, at 1100°C and 1 atm is fed to the converter; see the sketch below. (Hint: energy balances are not required). (a) Calculate flow rate of the product stream P in mol/hr and in m'/hr at 1500°C and 1 atm and its composition in mole percent. (b) Calculate the flow rate of the recycle stream R in mol/hr and in m'/hr at 1500°C and 1 atm. R (kg mol) 100% SO2 1500°C, 1 atm Separator P (kg mol) mol fr mol SO3 XSO3 O2 N2 nso3 no2 XN2 F(kg mol) 1.00 P G(kg mol) mol fr Soo 1.00 Converter 10°m/hr 1100°C A(kg mol) mol fr O2 0.21 N2 0.79 1.00 1 atm 25°C 1 atm
Expert Answer:
Answer rating: 100% (QA)
Solution By considering material balance around the SO 2 feeding point F R G Given F 10 6 m 3 hr By ... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these finance questions
-
When methanol, CH3OH,CH3OH, is burned in the presence of oxygen gas, O2,O2, a large amount of heat energy is released. For this reason, it is often used as a fuel in high performance racing cars. The...
-
A sulfuric acid plant produces a considerable amount of heat. This heat is used to generate electricity, which helps reduce operating costs. The synthesis of H2SO4 consists of three main chemical...
-
Propane fuel (C3H8) is burned in the presence of air. Assuming that the combustion is theoretical-that is, only nitrogen (N2), water vapor (H2O), and carbon dioxide (CO2) are present in the...
-
If there is a decrease in the demand for Canadian dollars relative to U.S. dollars, a. The price and quantity of Canadian dollars traded will fall. b. The price and quantity of Canadian dollars will...
-
Citigroup issued bonds that pay a 5.5 percent coupon interest rate. The bonds mature in 5 years. They are selling for $ 1,076. What would be your expected rate of return (yield to maturity) if you...
-
Refer to the previous problem. In 1984, Congress allowed G&H to sell mortgages at a loss and to amortize the loss over the remaining life of the mortgage. If this were used for the previous problem,...
-
Use your calculator to calculate \((-18) \times 13\). Explain how the answer agrees with what was expected.
-
The annual data that follow pertain to Flannery Water Optics, a manufacturer of swimming goggles (the company had no beginning inventories): Sales...
-
Last Chance Mine (LC) purchased a coal deposit for $780,000. Itestimated it would extract 12,000 tons of coal from the deposit. LCmined the coal and sold it reporting gross receipts of $1 millionfo 2...
-
The soil mass in Figure P9-13 is loaded by a force transmitted through a circular footing as shown. Determine the stresses in the soil. Compare the values of r using an axisymmetric model with the ...
-
vity: Sales Increase Maggie's Muffins, Inc., generated $4,000,000 in sales during 2015, and its year-end total assets were $2,400,000. Also, at year-end 2015, current liabilities were $1,000,000,...
-
Imagine that you are the manager of a boat manufacturing company and you are considering the purchase of new land in a year. The price of the land is $13,000. The company's capital structure is...
-
M10.4. A software installation project is being reviewed. Assume the project must be completed in 16 weeks. Crash the project as necessary (crashing one week at a time). The relevant information is...
-
Question 3. Tables. Reconstruct the sentence, table and caption shown below. To label the table, use \labelftab: Math2024tasks) after the caption line. To make the cross- reference to "Table 1" in...
-
When the gasoline pump shut off automatically, 23.6 gallons at $2.12 is the amount that will show on the cost dial? 129/10 per gallon had been delivered. What The cost dial will read $ (Round to the...
-
Q4: Show that tan 11/2 +tan12/11=tan 13/4 2 25: If A=[3], B=[ -1 31 find 2A-B 0
-
please solve this in java, dont use private class, just the public class. . A Ba gel Recipe can be represented as the amount of ?our, water, yeast, salt, and malt in the recipe. A perfect bagel res...
-
If the joint cost function for two products is C(x, y) = xy2 + 1 dollars (a) Find the marginal cost (function) with respect to x. (b) Find the marginal cost with respect to y.
-
Consider the manometer in Fig. P1-78. If the specific weight of fluid A is 100 kN/m3, what is the absolute pressure, in kPa, indicated by the manometer when the local atmospheric pressure is 90 kPa?
-
An air standard cycle with constant specific heats is executed in a closed piston-cylinder system and is composed of the following three processes: 1-2 Constant volume heat addition 2-3 lsentropic...
-
Reconsider Prob. 5 - 74. Using EES (or other) software, investigate the effect of the mass flow rate of the cold stream of R-134a on the temperature and the quality of the exit stream. Let the ratio...
-
(a) Why do some businesses keep a petty cash book as well as a cash book? (b) Kathryn Rochford keeps her petty cash book on the imprest system, the imprest being 25. For the month of April 19X9 her...
-
On 1 March 19X6, C Black, Curzon Road, Stockport, sold the following goods on credit to J Booth, 89 Andrew Lane, Stockport SK1 1AA, Order No 1697: (a) Prepare the sales invoice to be sent to J Booth....
-
On 1 May 19X7, D Wilson Ltd, 1 Hawk Green Road, Stockport, sold the following goods on credit to G Christie & Son, The Golf Shop, Hole-in-One Lane, Marple, Cheshire: Order No A/496 3 sets Boy Michael...

Study smarter with the SolutionInn App


