Complete the following questions based on the remote laboratory procedures: 1. What are some similarities in...
Fantastic news! We've Found the answer you've been seeking!
Question:
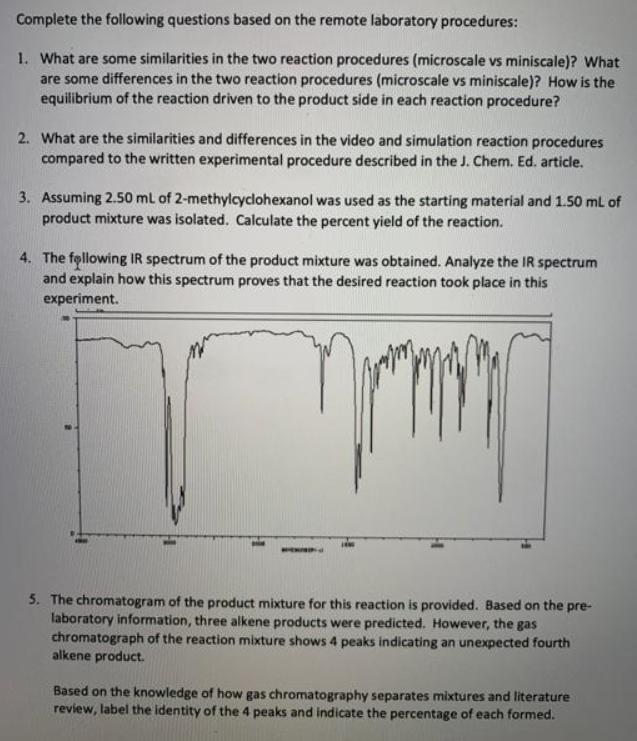
Transcribed Image Text:
Complete the following questions based on the remote laboratory procedures: 1. What are some similarities in the two reaction procedures (microscale vs miniscale)? What are some differences in the two reaction procedures (microscale vs miniscale)? How is the equilibrium of the reaction driven to the product side in each reaction procedure? 2. What are the similarities and differences in the video and simulation reaction procedures compared to the written experimental procedure described in the J. Chem. Ed. article. 3. Assuming 2.50 ml of 2-methylcyclohexanol was used as the starting material and 1.50 mL of product mixture was isolated. Calculate the percent yield of the reaction. 4. The following IR spectrum of the product mixture was obtained. Analyze the IR spectrum and explain how this spectrum proves that the desired reaction took place in this experiment. 5. The chromatogram of the product mixture for this reaction is provided. Based on the pre- laboratory information, three alkene products were predicted. However, the gas chromatograph of the reaction mixture shows 4 peaks indicating an unexpected fourth alkene product. Based on the knowledge of how gas chromatography separates mixtures and literature review, label the identity of the 4 peaks and indicate the percentage of each formed. Complete the following questions based on the remote laboratory procedures: 1. What are some similarities in the two reaction procedures (microscale vs miniscale)? What are some differences in the two reaction procedures (microscale vs miniscale)? How is the equilibrium of the reaction driven to the product side in each reaction procedure? 2. What are the similarities and differences in the video and simulation reaction procedures compared to the written experimental procedure described in the J. Chem. Ed. article. 3. Assuming 2.50 ml of 2-methylcyclohexanol was used as the starting material and 1.50 mL of product mixture was isolated. Calculate the percent yield of the reaction. 4. The following IR spectrum of the product mixture was obtained. Analyze the IR spectrum and explain how this spectrum proves that the desired reaction took place in this experiment. 5. The chromatogram of the product mixture for this reaction is provided. Based on the pre- laboratory information, three alkene products were predicted. However, the gas chromatograph of the reaction mixture shows 4 peaks indicating an unexpected fourth alkene product. Based on the knowledge of how gas chromatography separates mixtures and literature review, label the identity of the 4 peaks and indicate the percentage of each formed.
Expert Answer:
Answer rating: 100% (QA)
O Simitarities are iven below Borh reactios will do in laboratoy scale BoM reachion oeme in ytass eq... View the full answer

Related Book For 

Marketing The Core
ISBN: 978-0078028922
5th edition
Authors: Roger A. Kerin, Steven W. Hartley, William Rudelius
Posted Date:
Students also viewed these languages questions
-
What are some similarities in the world's financial systems?
-
Complete the following analogies. What are some similarities and differences between the two concepts involved in each? a. Depreciation is to plant assets as is to intangible assets . b. Depreciation...
-
What are some similarities and differences between skimming pricing, prestige pricing, and above-market pricing?
-
How many of the following could be valid JDBC URL formats for an imaginary driver named magic and a database named box? A. Only first B. Only second C. Only third D. first and second E. first and...
-
Describe the five essential improvement activities.
-
Does the function f(x) appear to be continuous on the interval 0 x 2? If not, what about on the interval 0 x 0.5? 1 f(x) 2 x
-
Wittco Company reports pretax GAAP income in 2020, its first year of operations, of \(\$ 100,000\). Temporary differences in the GAAP basis and tax basis of assets arose in 2020 from the following...
-
The financial statements of Lioi Steel Fabricators are shown below, both the actual results for 2010 and the projections for 2011. Free cash flow is expected to grow at a 6% rate after 2011. The...
-
Please provide answer to this question. Question 27 of 50. Mark and Carrie are married, and they will file a joint return. They both work full-time, and their 2020 income totaled $89,000, all from...
-
ZIM Inc. (ZIM) is a high-technology company that develops, designs, and manufactures telecommunications equipment. ZIM was founded in Year 5 by Dr. Alex Zimmer, the former assistant head of research...
-
Sherpa Manufacturing has the following income statement for 6,000 units: Sales $600,000 Variable costs 360,000 Contribution margin 240,000 Fixed costs 80,000 Net income $160,000 (a) At what sales...
-
A $10,000 bond is redeemable at par in 5 years. When the yield rate is j2 = 12%, the purchase price is $8527.98. What is the coupon rate?
-
11:30 PM Fri 6 Oct Submit Verilog Assignment #1 _ Gradescope.pdf LI 02.1 5 Points Consider a circuit model referred to as MOD1 whose interface is defined as shown below: B24 X1 MOD1 Y1 B1 78% Done
-
Keri Co . had sales for the year totaling $ 6 0 , 0 0 0 . Based on experience, management estimates 4 % of sales will be refunded to customers. Management also estimates $ 1 , 0 0 0 of inventory will...
-
Compare and contrast digital evidence and non-digital evidence. Provide three examples of how they are similar and three examples of how they are different. You must use the provided course materials...
-
Your plan to adopt a new medical health care database management system, and have tasked you to create a request for proposal (RFP). Different vendors will compete to build and provide the system to...
-
8. In a simple pendulum with no friction, a pendulum bob swings along the circular arc indicated in the figure below mechanical energy is conserved. As the pendulum swings from point B to point C,...
-
Sue Deliveau opened a software consulting firm that immediately paid $2,000 for a computer. Was this event a transaction for the business?
-
1. How has an understanding of consumer behavior helped Groupon grow from 400 subscribers in Chicago in 2008 to 60 million subscribers in 40 countries today? 2. What is the Groupon Promise? How does...
-
What methods would you suggest to assess the potential commercial success for the following new products? (a) A new, improved ketchup; (b) A three-dimensional television system that took the company...
-
(a) Why might a marketing researcher prefer to use secondary data rather than primary data in a study? (b) Why might the reverse be true?
-
In the income statement, Freight-In is (a) added to purchases. (c) added to sales. (b) subtracted from purchases. (d) subtracted from cost of goods sold.
-
A purchase invoice for \($1,200\) with credit terms 2/10, n/30, and a return of \($300\) received by the seller prior to payment, is paid within the discount period. A check should be sent for (a)...
-
What steps are used to post purchases returns and allowances from the general journal to the general ledger and accounts payable ledger?

Study smarter with the SolutionInn App


