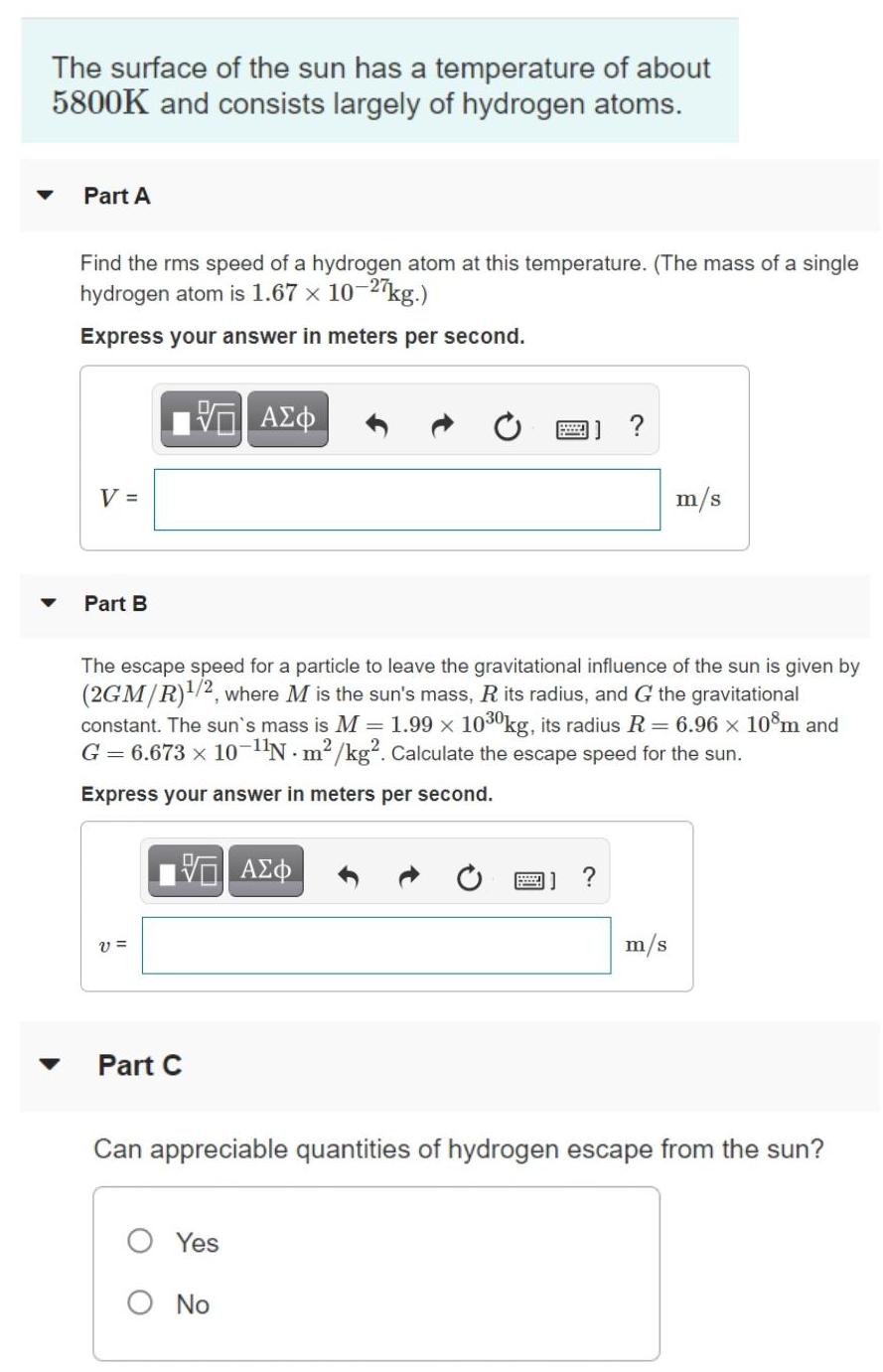
The surface of the sun has a temperature of about 5800K and consists largely of hydrogen...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
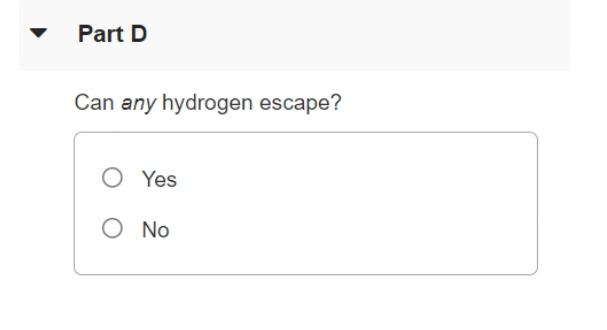
The surface of the sun has a temperature of about 5800K and consists largely of hydrogen atoms. Part A Find the rms speed of a hydrogen atom at this temperature. (The mass of a single hydrogen atom is 1.67 x 10-2 kg.) Express your answer in meters per second. V = m/s Part B The escape speed for a particle to leave the gravitational influence of the sun is given by (2GM/R)/2, where M is the sun's mass, R its radius, and G the gravitational constant. The sun's mass is M = 1.99 x 1030kg, its radius R = 6.96 x 10m and G = 6.673 x 10-N m2 /kg². Calculate the escape speed for the sun. Express your answer in meters per second. A) ? v = m/s Part C Can appreciable quantities of hydrogen escape from the sun? O Yes O No Part D Can any hydrogen escape? O Yes O No The surface of the sun has a temperature of about 5800K and consists largely of hydrogen atoms. Part A Find the rms speed of a hydrogen atom at this temperature. (The mass of a single hydrogen atom is 1.67 x 10-2 kg.) Express your answer in meters per second. V = m/s Part B The escape speed for a particle to leave the gravitational influence of the sun is given by (2GM/R)/2, where M is the sun's mass, R its radius, and G the gravitational constant. The sun's mass is M = 1.99 x 1030kg, its radius R = 6.96 x 10m and G = 6.673 x 10-N m2 /kg². Calculate the escape speed for the sun. Express your answer in meters per second. A) ? v = m/s Part C Can appreciable quantities of hydrogen escape from the sun? O Yes O No Part D Can any hydrogen escape? O Yes O No
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these physics questions
-
The surface of the Sun has a temperature of about 5 800 K. The radius of the Sun is 6.96 X 108 m. Calculate the total energy radiated by the Sun each second. Assume that the emissivity of the Sun is...
-
The surface of the Sun has a temperature of 5500 C and the temperature of deep space is 3.0 K. (a) Find the entropy increase produced by the Sun in one day, given that it radiates heat at the rate of...
-
The surface of the Sun has a temperature of about 6000 K. What is the speed of a typical hydrogen molecule in the Suns atmosphere?
-
Assume that Jimmy Cash has $2000 in his checking account at Folsom Bank and uses his checking account card to withdraw $200 of cash from the banks ATM machine. By what dollar amount did the M1 money...
-
The South Beach Cafe recently reduced appetizer prices from $12 to $10 for afternoon early bird customers and enjoyed a resulting increase in sales from 90 to 150 orders per day. Beverage sales also...
-
Graph the equation. Graph and interpret applications of slope-intercept. Costa is planning a lunch banquet. The equation \(C=450+28 g\) models the relation between the cost in dollars, \(C\), of the...
-
Newton Electronics Ltd has incurred expenditure of 5 million over the past three years researching and developing a miniature hearing aid. The hearing aid is now fully developed. The directors are...
-
According to the Pew report, 14.6% of newly married couples in 2008 reported that their spouse was of another race or ethnicity (CNNLiving, June 7, 2010). In a similar survey in 1980, only 6.8% of...
-
The General Social Survey has asked subjects, How long have you lived in the city, town, or community where you live now? The responses of 1415 subjects in one survey had a mode of less than 1 year,...
-
The chemical formula of feldspar is (A) KAISI3OS (B) Naz AlF, (C) Na AIO2 (D) K2SO4. Al (SOA)3-4Al(OH),
-
Mandeville Limited ( ML ) constructed a distillery on January 1 , 2 0 2 1 . The distillery is estimatedto have a useful life of 3 0 years ( nil residual value ) from the date it was completed and...
-
The assembly department of Whitby Inc. uses the weighted average method of process costing. They shared the following data for the quarter with you. Physical units Direct materials Conversion costs...
-
Examine the shift in accounting standards for loan loss provisioning from incurred loss models to expected credit loss models in the aftermath of the 2 0 0 7 - 0 8 Financial Crisis. How do the...
-
In another water circuit, water flows at a rate of 30 litres/minute through a Perspex pipe of external diameter 80mm. Given that the pipe has a thickness of 0.01mm and the dynamic viscosity of the...
-
When car has a lifetime distribution exp(1/4), the owner wants to change the car either it is broken or the car is 8 years old. If it is replaced at 8 years (not broken), the owner pays 20, 000...
-
Lamp Light Limited (LLL) manufactures lampshades. It applies variable overhead on the basis of direct labor hours. Information from LLL's standard cost card follows: Standard Standard Standard...
-
In Problems 718, write the augmented matrix of the given system of equations. f0.01x0.03y = 0.06 [0.13x + 0.10y = 0.20
-
Suppose a neuron in the brain carries a current of 5.0 10-8 A. Treating the neuron as a straight wire, what is the magnetic field it produces at a distance of 7.5 cm? A. 1.3 10-13 T B. 4.2 10-12 T...
-
Suppose you pour water into a container until it reaches a depth of 14 cm. Next, you carefully pour in a 7.5-cm thickness of olive oil so that it floats on top of the water. What is the pressure at...
-
Figure 28-49 shows a single-slit diffraction pattern formed by light passing through a slit of width W = 11.2 mm and illuminating a screen 0.855 m behind the slit. (a) What is the wavelength of the...
-
Railway Oil and Gas Company owned the following unproved property as of the end of 2010: Although no activity took place on Lease A during the year, Railway decided that Lease A was not impaired,...
-
Latitude Energy decided to explore some acreage in Texas before acquiring any leases. Latitude acquired shooting rights only on 15,000 acres owned by Mr. T for \($0.10/acre. Latitude\) obtained...
-
Sauer Energy purchased land in fee for $100,000. The fair market values of the surface and mineral rights were determined by a qualified appraiser as follows: Prepare a journal entry to record the...

Study smarter with the SolutionInn App


