1. a. Which of the following components has the highest vapor pressure? b. Which of the...
Fantastic news! We've Found the answer you've been seeking!
Question:
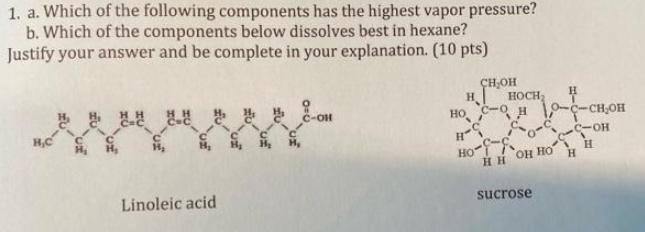
Transcribed Image Text:
1. a. Which of the following components has the highest vapor pressure? b. Which of the components below dissolves best in hexane? Justify your answer and be complete in your explanation. (10 pts) H,C Linoleic acid C-OH CH₂OH H HOCH₂ H но с-о 10-C-CH₂OH H-S -С-он Ho H HH OH HO sucrose H 1. a. Which of the following components has the highest vapor pressure? b. Which of the components below dissolves best in hexane? Justify your answer and be complete in your explanation. (10 pts) H,C Linoleic acid C-OH CH₂OH H HOCH₂ H но с-о 10-C-CH₂OH H-S -С-он Ho H HH OH HO sucrose H
Expert Answer:
Answer rating: 100% (QA)
Answer Linoleic acid has the highest vapor pressure of the two components This is because it is the lightest and has the lowest boiling point Sucrose ... View the full answer

Related Book For 

Posted Date:
Students also viewed these physics questions
-
A small particle of mass m is pulled to the top of a frictionless half-cylinder (of radius R) by a cord that passes over the top of the cylinder, as illustrated in Figure P7.20. (a) If the particle...
-
A small particle of mass m is released from rest in a viscous fluid to fall under gravity. The particle experiences a linear drag force F = -bv, measuring distance z downwards as the positive...
-
A particular particle of mass m is in an infinite well of width L. The particle is in the n = 3 energy level. What is the energy of the particle in this state? Show that this energy is also the...
-
In Exercises determine whether Rolle's Theorem can be applied to on the closed interval [a, b]. If Rolle's Theorem can be applied, find all values of c in the open interval (a, b) such that '(c) =...
-
1. Andersons bank requires a compensating balance of $3 million. How much additional funds can be freed up for investment in fixed assets if the firm reduces its cash balance to the minimum required...
-
Verify that the function corresponding to the following figure is a valid probability density function. Then find the following probabilities: a. P(x < 8) b. P(x < 8) c. P(6 < x < 10) d. P(6 < x <...
-
In the chapter we have: If the firm has a low retention rate and a large growth rate what does this imply? Would you expect a high or low P/E ratio? P/E = (1-b) k-g
-
Waterworld Company leased equipment from Costner Company. The lease term is 4 years and requires equal rental payments of $43,019 at the beginning of each year. The equipment has a fair value at the...
-
Q3: Find two positive numbers x and 3.! whose sum is 30 and the product of one of them by the square ofthe other is as great as possible
-
1. Assume a 30% tax rate, and the Totals per financial statements provided. Complete the following schedule as per Figure 16.4. Assume that the Totals per financial statements (second to bottom row)...
-
! Required information [ The following information applies to the questions displayed below. ] Melissa recently paid $ 5 9 5 for round - trip airfare to San Francisco to attend a business conference...
-
Part 1. Determine the function of the underlined asset in each of the following situations. (2 points each) 1. Shaira uses cash to buy school supplies for her son. 2. After having her dinner, Moira...
-
How can an organization ensure that they are asking the right questions during the market research process?
-
Basket Company specializes in unique baskets. Peak sales for one of their products, the Easter basket, occur in March every year. The company has estimated the following sales for the first five...
-
Comparative Ratios. (LO10-4) The government-wide financial statements for the City of Arborland for three years are presented below. Additional information follows: Population: Year 2023: 28,420,...
-
hat ways does the structure of an organization influence its effectiveness, and which organizational designs are best suited for promoting adaptability and innovation ?
-
Follow the directions to verify the identity Do not solve as an equation sec x tan x COS X 1 sinx Directions Start from the right Multiply the fraction top and down by 1 sin x Simplify and or rewrite...
-
The Zwatch Company manufactures trendy, high-quality moderately priced watches. As Zwatch's senior financial analyst, you are asked to recommend a method of inventory costing. The CFO will use your...
-
How much energy can you store on a parallel plate capacitor with d = 1 m, A = 10 cm 2 , and = 100 0 ,assuming that the breakdown field of the dielectric is the same as for air?
-
A geothermal resource outputs saturated (liquid) water at 250 C (as in Example 32.5). Consider two alternative ways to use this resource to deliver space heating at T = 20 C. First, the hot water...
-
One way to estimate the effective area of an object is to measure its limiting velocity v falling in air. Explain how this works and find the expression for A eff as a function of m (the mass of the...
-
Match the descriptions on the left with the forms of business enterprise on the right: 1. Most numerous 2. Commands most revenues 3. Two or more co-owners 4. Has stockholders 5. Owned by only one...
-
Use the accounting equation to answer each question below. 1. The assets of Tiller Company are $240,000, and the liabilities are $180,000. What is the amount of the stockholders equity? 2. The...
-
Carlton Company had assets of $280,000 and liabilities of $120,000 at the beginning of the year, and assets of $400,000 and liabilities of $140,000 at the end of the year. During the year, there was...

Study smarter with the SolutionInn App


