The following liquid-phase reactions were carried out in a CSTR at 325 K. kLA =...
Fantastic news! We've Found the answer you've been seeking!
Question:
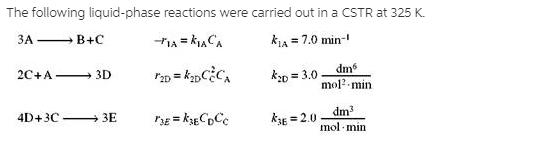

Transcribed Image Text:
The following liquid-phase reactions were carried out in a CSTR at 325 K. ЗА kLA = 7.0 min- - B+C k0 = 3.0 dm mol?-min 2C+A- 3D dm mol · min 4D+3C 3E kE = 2.0 !! The concentrations measured inside the reactor were CA = 0.10, CB = 0.93, CC = 0.51 , and CD = 0.049 all in mol/dm3. %3D (a) What are 1A, 12A, and 13A? (1A = - 0.7 mol/dm3 • min) (b) What are 1B , 2B , and r3B? (c) What are 1C, r2C, and r3C? (1C = 0.23 mol/dm3 •min) (d) What are r1D, 12D , and 13D? (e) What are 1E• 2E, and r3E? (f) What are the net rates of formation of A, B, C, D, and E? (g) The entering volumetric flow rate is 100 dm3/min and the entering concentration of A is 3M. What is the CSTR reactor volume? (h) Write a Polymath program to calculate the exit concentrations when the volume is given as 600 dm3 (1) PFR. Now assume the reactions take place in the gas phase. Use the preceding data to plot the molar flow rates as a function of PFR volume. The pressure drop parameter is 0.001 dm-3, the total concentration entering the reactor is 0.2 mol/dm3, and uo = 100 dm3/min. What are ŚD/E and SC/D? () Membrane Reactor. Repeat (i) when species C diffuses out of a membrane reactor and the transport coefficient, kC, is 10 min-1. Compare your results with part (1). The following liquid-phase reactions were carried out in a CSTR at 325 K. ЗА kLA = 7.0 min- - B+C k0 = 3.0 dm mol?-min 2C+A- 3D dm mol · min 4D+3C 3E kE = 2.0 !! The concentrations measured inside the reactor were CA = 0.10, CB = 0.93, CC = 0.51 , and CD = 0.049 all in mol/dm3. %3D (a) What are 1A, 12A, and 13A? (1A = - 0.7 mol/dm3 • min) (b) What are 1B , 2B , and r3B? (c) What are 1C, r2C, and r3C? (1C = 0.23 mol/dm3 •min) (d) What are r1D, 12D , and 13D? (e) What are 1E• 2E, and r3E? (f) What are the net rates of formation of A, B, C, D, and E? (g) The entering volumetric flow rate is 100 dm3/min and the entering concentration of A is 3M. What is the CSTR reactor volume? (h) Write a Polymath program to calculate the exit concentrations when the volume is given as 600 dm3 (1) PFR. Now assume the reactions take place in the gas phase. Use the preceding data to plot the molar flow rates as a function of PFR volume. The pressure drop parameter is 0.001 dm-3, the total concentration entering the reactor is 0.2 mol/dm3, and uo = 100 dm3/min. What are ŚD/E and SC/D? () Membrane Reactor. Repeat (i) when species C diffuses out of a membrane reactor and the transport coefficient, kC, is 10 min-1. Compare your results with part (1).
Expert Answer:

Related Book For 

Applied Regression Analysis and Other Multivariable Methods
ISBN: 978-1285051086
5th edition
Authors: David G. Kleinbaum, Lawrence L. Kupper, Azhar Nizam, Eli S. Rosenberg
Posted Date:
Students also viewed these chemistry questions
-
The following liquid-phase reactions were carried out in a CSTR at 325 K. 3A B+C -rIA = KIACA kIA = 6.0 min-ls dm mol2 min 2C+A 3D k2p = 4.0 dm3 dm3 4D+3C- 3E r3E = k3E CpCc k3E = 5.0 mol min The...
-
A five-year follow-up study was carried out in a certain metropolitan area to assess the relationship of diet and weight to the incidence of stomach cancer. Data were obtained on n = 2,000 subjects....
-
The following reaction is carried out in a 10-L aqueous solution in a stirred tank A + B --> AB At the outset of the reaction the reagents A and B are added to the reactor in equal 1-mole...
-
Ratio Computation and Analysis; Liquidity) as loan analyst for Madison Bank, you have been presented the following information. Each of these companies has requested a loan of $50,000 for 6 months...
-
Computer control of a robot to spray-paint an automobile is accomplished by the system shown in Figure DP5.8(a) [7]. We wish to investigate the system when K = 1, 10, and 20. The feedback control...
-
Convert each of the following using unit conversion factors show the cancellation of the units Convert each of the following using the definitions in Table DI.I and unit conversion factors. Show the...
-
U.S. Airways recognizes revenue from airline ticket sales when a customer takes a flight or not later than one year from the date of sale (flight coupons have a useful life of one year from the date...
-
Morris Gray leased waterfront property on the Ross Barnett Reservoir to a restaurant, Edgewater Landing, Inc., for a 10-year term. After a year and a half, Edgewater's original shareholder, Billy...
-
Smithson Mining operates a silver mine in Nevada. Acquisition, exploration, and development costs totaled $6.3 million. After the silve is extracted in approximately five years, Smithson is obligated...
-
Hannah Turnbull manages Elegant Suites, a hotel in a small town 10 miles inland from Florida's beautiful gulf coast. Elegant Suites has a capacity of 320 suites and offers a small, but well managed,...
-
For items 3-7, use the following facts: Reiner, Annie and Bertolt formed a partnership while doing their mission in the Paradis Island with a profit and loss ratio of 2:3:5. Annie fell in love with...
-
Assume that the government is unable to change the amount of public debt that the private sector holds. Consider both fixed and flexible exchange rate regimes. Explain how would the nominal and real...
-
Assume that you have been invited to ONE of the following social/cultural events for the very first time: 1.a Jewish Passover Seder 2.a Day of the Dead family picnic in a Mexican graveyard 3.a formal...
-
Explain the difference between minutes of meetings and reports of outcomes of meetings.?
-
Add. 4 2-2x + x-1 X Simplify your answer as much as possible.
-
The mass of a large concrete block is 5 5 8 kg . Is this a scalar quanity? explain
-
#5 Create an appropriate figure to examine the distribution of age in the sample (make sure both axes titles are in Title Case!). Number it Figure 1 and create an appropriate title. In the Note...
-
Under what conditions is the following SQL statement valid?
-
A study by Rikkers et al. (1978) involved a prospective randomized surgical trial that compared cirrhotic patients who had bled from use of either a nonselective shunt (a standard operation) or a...
-
a.-e. Repeat Problem 8, parts (a) through (e), after centering the predictor (age). f. Compare the results obtained here to those obtained in Problem 8. Problem 8 This problem uses the data presented...
-
The accompanying source table relates to a study involving the effects of trimethylin doses of 0, 3, 6, and 9 mg/kg on a sample of 48 rats. Each rat received only one dose. The response variable was...
-
Handy Dan, Inc., operates warehouse-style stores, selling a variety of home building products and lawn and garden supplies. Presented below are Handy Dans historical financial statements for 2014 and...
-
Presented below are profitability ratios for three competitors: Bristol-Myers Squibb (BMY), Pfizer, Inc. (PFE), and Johnson & Johnson (JNJ): Required Compare the relative profitability of the three...
-
Presented below are selected financial statement data for three global energy firms: Required 1. Compare the three firms in terms of their size and their financing strategy. 2. Evaluate the...

Study smarter with the SolutionInn App


