Write out half equations, the cell reaction, and determine the standard potential of the following cells given
Question:
Write out half equations, the cell reaction, and determine the standard potential of the following cells given standard electrode potentials
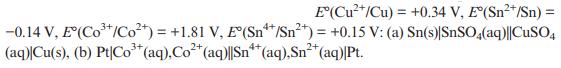
Transcribed Image Text:
E(Cu+/Cu) = +0.34 V, E(Sn*/Sn) = -0.14 V, E(Co*/Co) = +1.81 V, E(Sn/Sn+) = +0.15 V: (a) Sn(s)|SnSO4(aq)||CuSO4 3+ 2+ (aq)|Cu(s), (b) Pt|Co+ (aq), Co+ (aq)|Sn** (aq), Sn+ (aq)|Pt.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (7 reviews)
a Cu 2 2e Cu E 034 V higher standard potential defines the oxidizing agent Sn 2 2e ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
As in Example 6L.1, you are planning to use a Daniell cell to power a model electric car. However, you find that you do not have standard solutions available. You have only dilute solutions, and you...
-
Write the cell reaction and electrode half-reactions and calculate the standard emf of each the following cells: (a) Ptl C12 (g) I HCl (aq) 11 K, Cr04 (aq) IAg, Cr04(s) IAg (b) Pt 1 Fe3+(aq),Fe2+(aq)...
-
The list below gives the standard electrode potentials for five half-reactions. Cu 2+ (aq) + e Cu(s) E = +0.34 V Fe 2+ (aq) + 2e Fe(s) E = 0.44 V Fe 3+ (aq) + e Fe2+(aq) E = +0.77 V I 2 (aq)...
-
Blackmon Company provides locator services to the city transportation departments. Blackmons service involves installing a dedicated hardware transmitter in each city bus. This transmitter provides...
-
Determine the wavelengths of spectral lines appearing on transition of excited Li atoms from the state 3S down to the ground state 2S. The Rydberg corrections for the S and P terms are 0.41 and ...
-
The lift sling is used to hoist a container having a mass of 500 kg. Determine the force in each of the cables AB and AC as a function of . If the maximum tension allowed in each cable is 5 kN,...
-
What do the following terms mean? (a) term loan, (b) bonds payable, (c) trustee, (d) secured bonds, (e) serial bonds, (f) call provision, (g) convertible bonds, (h) face value, (i) coupon rate, (j)...
-
During the current year, Zeta Corporation distributes the assets listed below to its sole shareholder, Susan. For each asset listed, determine the gross income recognized by Susan, her basis in the...
-
4. Outline and explain predictive models developed. Outline prediction models developed. There should be at least six different SVM models developed. Vary the appropriate parameters of the models...
-
In its charged state, a leadacid battery contains PbO 2 and Pb electrodes. The half equations that occur during discharge can be written as below. Give the overall cell equation and estimate E cell....
-
What is unusual about the superconducting phases Nd 2x Ce x CuO 4 and Sr 1x Nd x CuO 2 ?
-
Why must we restrict the domain of a quadratic function when finding its inverse?
-
When 0 . 3 mole of O 2 mixed with 0 . 5 mole N 2 , the total pressure was 0 . 2 atm. Calculate the partial pressure of N 2
-
A -6.80nC charge of mass 4.40E-6 kg is released from rest 4.10mm above a very large plane of positive charge. The charge accelerates toward the plane and collides with a speed of 4.50m/s. What is the...
-
At the end of the year 2004 the BRK Corporation had free cash flow to equity (FCFE) of $250,000 and shares outstanding of 200,000. The company projects the following annual growth rates in FCFE Year...
-
The temperature dependence of the resistivity of copper and copper-nickel alloys is shown in the figure below. Explain the reason why the resistivity is increased with Ni amount. What is the residual...
-
The moon is 3.8 x 10 km away frein the earth. How long a time will pass between the instant an astronaut on the moon speaks and the instant his veice is heard on earth? (His voice travels by laser...
-
Blue Sea Health Spas began 2012 with cash of $112,000. During the year, Blue Sea earned service revenue of $599,000 and collected $593,000 from customers. Expenses for the year totaled $380,000, of...
-
Suppose that you could invest in the following projects but have only $30,000 to invest. How would you make your decision and which projects would you invest in? Project Cost $ 8,000 11,000 9,000...
-
Compound A has molecular formula C 5 H 11 Br. When compound A is treated with bromine in the presence of UV light, the major product is 2,2-dibromopentane. Treatmentof compound A with NaSH (a strong...
-
In calculating H o R at 285.15 K, only the H o f of the compounds that take part in the reactions listed in Tables 4.1 and 4.2 (Appendix B, Data Tables) are needed. Is this statement also true if you...
-
Predict the products when each of the following compounds is treated with NBS and irradiated with UV light: (a) (b) (c) (d)
-
Use the given stock table to find the closing price for ABC Technologies (ABC). 52-Week Net Div High Low Stock 70.34 42.73 ABCTch SYM Amt ABC 3.00 Yld % PE 5.5 17 Vol 100s Close Chg 5915 54.58 1.10...
-
The Normal Distribution Make sure you answer all questions in context and print out any outputs and graphs that you perform in RStudio. These printouts will serve as justification for your work. For...
-
say you have a loan of 1 0 0 , 0 0 0 with a 3 0 year fixed rate, 6 . 8 8 % intrest, APR of 6 . 9 2 % you pay a total of 2 6 5 , 0 8 2 . 4 0 over the entire length of the loan ( 3 0 yrs ) . Suppose...

Study smarter with the SolutionInn App


