Prove analytically that the entropy of a two-statesystem? is maximized when p + = p ? =
Question:
Prove analytically that the entropy of a two-statesystem?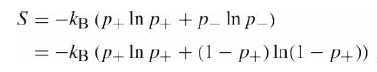
is maximized when p+ = p? = 1/2.
Transcribed Image Text:
S = -kB (p+ In p+ +p_ In p_) = -kB (P+ In p4 + (1 – P+) In(1 – P+))
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
Let p x then S k B x ln x 1 x ln1 x so at an ex...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
Prove analytically that the convolution of any function (x) with a delta function, (x), generates the original function (X).
-
Show that radiation at a distribution of temperatures decreases the estimate (34.34) of the uniform temperature radiative response 0 by proving the result in the case where the radiation comes from...
-
The third law of thermodynamics states that the entropy of a perfect crystal at 0 K is zero. In Appendix 4, F-(aq), OH-(aq), and S2-(aq) all have negative standard entropy values. How can S8 values...
-
Derek and Wes take a photograph of a lake at the same angle and the same time of day. Derek's picture comes out with crisp edges around objects in the photo. Wes's picture seems to blur the outlines...
-
Before attempting this problem, review Examples 7 and 8. Two curves on a highway have the same radii. However, one is unbanked and the other is banked at an angle u. A car can safely travel along the...
-
Action Athletics manufactures sporting goods that are then sold to retailers. It is a very competitive industry, where quality and price are important to gain space on retailers shelves. Actions...
-
Look at each of the following employees of Agetros Restaurant. Which of the elements of the fraud triangle apply? Perceived pressure (P) Perceived opportunity (O) Rationalization (R) 1. As the...
-
This year, Sigma Inc. generated $612,000 income from its routine business operations. In addition, the corporation sold the following assets, all of which were held for more than 12 months. a....
-
Plantation Green Limited is a major producer of processed coffee. On the 17 July 2018, it agreed to sell 200 kilograms of coffee to Grey-Drinks Restaurant. Both parties are located in X country in...
-
Indianapolis Pharmaceuticals Company (IPC) recently revised its performance evaluation system. The company identified four major goals and several objectives required to meet each goal. Kris...
-
Consider an amount of helium gas at atmospheric pressure and room temperature (T = 300K) enclosed in a 1-liter partition within a cubic meter. The volume outside the partition containing the helium...
-
Consider air to be a mixture of 78% nitrogen, 21%oxygen, and 1% argon. Estimate the minimum amount of energy that it takes to separate a cubic meter of air into its constituents at STP, by computing...
-
Write Lewis structures for the following molecules: (a) ICl, (b) PH3, (c) P4 (each P is bonded to three other P atoms), (d) H2S, (e) N2H4, (f) HClO3, (g) COBr2 (C is bonded to O and Br atoms).
-
Dalton Manufacturing is preparing its master budget for the first quarter of the upcoming year. The following data pertain to Dalton Manufacturing's operations: (Click the icon to view the data.)...
-
11. Let A= -237---8- and = -H] Suppose and v are eigenvectors of A. Use this information to diagonalize A.
-
"Feistels cipher (software implementation with 2 ciphering modes ECB and CBC)" show how feistels cipher implements and use 7 test cases to prove DOCUMENTATION: Description of the used algorithm...
-
Advise me how I use the following information to fill out a form 1120? ( For Form 1120, Schedule K (on pages 4 and 5), you only need to complete lines 1 - 2 (a, b & c). You can disregard the...
-
Lynch Company manufactures and sells a single product. The following costs were incurred during the company's first year of operations: Variable costs per unit: Manufacturing: Direct materials Direct...
-
Amanda M. worked the following schedule: Monday, 8 hours; Tuesday, 9 hours; Wednesday, 7 hours 48 minutes; Thursday, 8 hours; Friday, 8 hours. The employer pays overtime for all time worked in excess...
-
The following selected accounts and normal balances existed at year-end. Notice that expenses exceed revenue in this period. Make the four journal entries required to close the books: Accounts...
-
If a pilot accelerates at more than 4g, he begins to gray out but doesnt completely lose consciousness. (a) Assuming constant acceleration, what is the shortest time that a jet pilot starting from...
-
Air-Bag Injuries. During an auto accident, the vehicles air bags deploy and slow down the passengers more gently than if they had hit the windshield or steering wheel. According to safety standards,...
-
Prevention of Hip Fractures. Falls resulting in hip fractures are a major cause of injury and even death to the elderly. Typically, the hips speed at impact is about 2.0 m/s. If this can be reduced...
-
k Two grams of musk oil are required for each bottle of Mink Caress, a popular perfume made by a company in western Siberia. The cost of the musk oil is $1.60 per gram. Budgeted quarterly production...
-
1.Explainthebasicdifferencesbetweentheoperationofacurrencyforwardmarketandafuturesmarket. 2.Assume today's settlement price on a CME, EUR futures contract is $1.3140/EUR. You haveashortposition...
-
1. Communication and the concept of "communicative intelligence" are introduced in ELM #1 as "the lifeblood of [an] organization." This means that effective professional communication is an essential...

Study smarter with the SolutionInn App


