Verify the expressions for the Q-value in ? ? -decay and electron capture, eq. (17.34)? Q(B) =
Question:
Verify the expressions for the Q-value in ??-decay and electron capture, eq. (17.34)?
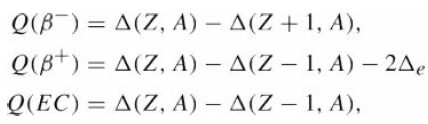
Transcribed Image Text:
Q(B¯) = A(Z, A) – A(Z + 1, A), Q(B+) = A(Z, A) – A(Z – 1, A) – 2Ae Q(EC) = A(Z, A) - A(Z – 1, A),
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
For decay eq 1732 gives adding and subtrac...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
Use Eq. (9.124) of Prob. 9.14 to verify the expressions for the integrals (11| 22) and |22| 22) in the Section 14.3 example In Eq. 9.14 E = E) + AE) + xE2)
-
In the Section 16.2 CI example, verify the expressions given in terms of integrals over the basis functions for (a) (b) (D2|2);
-
In both internal conversion and decay and electron is emitted. How conversion and decay, an electron is emitted. How could you determine which decay process occurred?
-
Crosby is known for his advocacy of zero-defects management and prevention; Crosby's Quality Vaccine consists of which three ingredients? Group of answer choices A. Determination, Education, and...
-
A Boeing 747 "Jumbo Jet" has a length of 59.7 m. The runway on which the plane lands intersects another runway. The width of the intersection is 25.0 m. The plane decelerates through the intersection...
-
Barton Company is contemplating the acquisition of the net assets of Crowley Company for $850,000 cash. To complete the transaction, acquisition costs are $15,000. The balance sheet of Crowley...
-
Speegleville Marina needs to raise \(\$ 1\) million to expand. Speegleville's president is considering two plans: - Plan A: Issue \(\$ 1,000,000\) of \(8 \%\) bonds payable to borrow the money - Plan...
-
Hammer Orthopedic Corporation periodically invests large sums in marketable equity securities. The investment policy is established by the investment committee of the board of directors and the...
-
The owner of Britten's Egg Farm wants to estimate the mean number of eggs laid per chicken. A sample of 16 chickens shows they laid an average of 18 eggs per month with a standard deviation of 5 eggs...
-
Last month the Henke Company had sales ot $220,000, a C/M ratio of 40%, and an M/S ratio 30%. During the current month, a decrease in sales price and a decrease in fixed costs have resulted in a C/M...
-
Check that 3 He is less tightly bound than 3 H. Explain why 3 H decays to 3 He, and not vice versa.
-
Ignoring small electron binding energies and the very small mass of the neutrino, show that the mass of a nucleus increases when it decays by electron capture if the Q-value of the decay is less than...
-
Do U.S. GAAP and IFRS differ in the amount of flexibility that companies have in electing the fair value option? Explain.
-
how does Innocent Drinks understands sustainable and ethical marketing and incorporates this into their marketing strategy; consider why it is done in this way.
-
Explain the importance of crisis management and adaptability in an organization, and in particular for a leader. How are crisis management and adaptability essential aspects of leadership? What type...
-
In own words, explain the process used in selection and implementation of an EHR. Report on project management and strategic planning methodologies and how the functions of both support the...
-
A 4x4 meter [45/-45]s balanced symmetric layup (note 4 plies) is used in a space applications where the change in temperature is AT=-100C. Once on orbit at this temperature, the sample experiences...
-
Multiply. (5x)(-4x) (3)
-
How are secondary sources on U.S. GAAP used to help the researcher?
-
(a) Given a mean free path = 0.4 nm and a mean speed vav = 1.17 105 m/s for the current flow in copper at a temperature of 300 K, calculate the classical value for the resistivity of copper. (b)...
-
A 20.0-L tank contains 4.86 10 4 of helium at 18.0 o C. The molar mass of helium is 4.00 g/mol. (a) How many moles of helium are in the tank? (b) What is the pressure in the tank, in pascals and in...
-
Helium gas with a volume of 2.60 L, under a pressure of 0.180 atm and at a temperature of 41.0 o C, is warmed until both pressure and volume are doubled. a. What is the final temperature? b. How many...
-
A cylindrical tank has a tight-fitting piston that allows the volume of the tank to be changed. The tank originally contains 0.110 m 3 of air at a pressure of 0.355 atm. The piston is slowly pulled...
-
Arizona Corporation acquired the business Data Systems for $315,000 cash and assumed all liabilities at the date of purchase. Data's books showed tangible assets of $330,000, liabilities of $18,000,...
-
Define and describe the concepts of IMC (Integrated Marketing Communications) and RTM (real time marketing). How does this relate to the discussion in the textbook of time compression and online vs....
-
Activity The Serada IT is installing a new computer system. Several thing must be done to make sure the system is working properly. A list of activities of optimistic, most likely and pessimistic...

Study smarter with the SolutionInn App


