a. Name each of the following alcohols. b. Name each of the following alcohols, including the stereochemistry
Question:
a. Name each of the following alcohols.
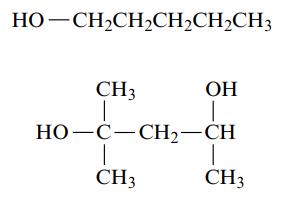
b. Name each of the following alcohols, including the stereochemistry if cis–trans isomers are possible.
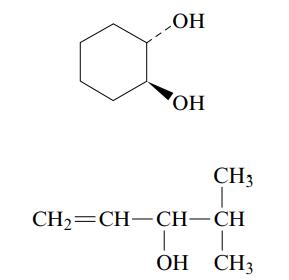
Transcribed Image Text:
HỌ—CH,CH,CH,CH,CH3 ОН I CH3 T HỌ–C–CH2–CH 1 CH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
ANSWER Alcohols are substances in which any carbon atom in the main carbon chain has a hydroxyl grou...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Each of the following alcohols has been subjected to acidcatalyzed dehydration and yields a mixture of two isomeric alkenes. Identify the two alkenes in each case, and predict which one is the major...
-
Each of the following alcohols has been prepared by reaction of a Grignard reagent with ethylene oxide. Select the appropriate Grignard reagent in each case. (a) CH3 CH2CH2OH CH2CH20
-
Each of the following alcohols has been subjected to acidcatalyzed dehydration and yields a mixture of two isomeric alkenes. Identify the two alkenes in each case, and predict which one is the major...
-
Bruno Corp. has decided to expand its operations. The bookkeeper recently completed the following statement of financial position in order to obtain additional funds for expansion: Instructions (a)...
-
The Hughes Supply Company uses an inventory management method to determine the monthly demands for various products. The demand values for the last 12 months of each product have been recorded and...
-
Use mathematical induction to prove that for n > 2, ..+ an-12"-1 +1") det(C, A1) = (-1)"(o + ajd + = (-1)" p(2)
-
Every day Elise looks at the bond section of The Wall Street Journal to see if she can get a good deal on a $1,000 bond and add it to her portfolio. This morning a price quotation of 98 for...
-
Victoria Companys balance sheet at December 31, 2013, is presented below. During January 2014, the following transactions occurred. Victoria uses the perpetual inventory method. Jan. 1 Victoria...
-
How do I calculate this? Karen bought a car 3 years ago for $16000 and ?nanced the full purchase price with a 3 year (36 month) simple interest car loan at 5% interest (monthly payment = $479.53)....
-
It's mid-December and Marie is finalizing her divorce. Generally, she and her soon to be ex-spouse have been amicable, and Marie is looking forward to being single. The divorce attorneys will be...
-
Estradiol is a female hormone with the following structure: How many chiral carbon atoms are in estradiol? HO CH3 OH
-
The two isomers having the formula C 2 H 6 O boil at -23 C and 78.5 C. Draw the structure of the isomer that boils at -23 C and of the isomer that boils at 78.5 C.
-
A distillate containing 45 wt% isopropyl alcohol, 50 wt% diisopropyl ether, and 5 wt% water is obtained from the heads column of an isopropyl alcohol finishing umt. The company desires to recover the...
-
Briefly explain the nature of methods standards, performance standards, and documentation standards.
-
Why are managers so necessary in organizations?
-
a. A supervisor is well advised to treat, as much as possible, all members of his/her group exactly the same way. b. A supervisor is well advised to adjust his/her behavior according to the unique...
-
Briefly explain three factors that are likely to affect whether a transfer pricing scheme for information systems services is likely to be successful as a control mechanism.
-
a. Generally speaking, individual motivation is greatest if the person has set goals for himself/herself that are difficult to achieve. b. Generally speaking, individual motivation is greatest if the...
-
Use the following Russian (Russian Federation) balance of payments data from the IMF to answer questions 3.14 through 3.17. 1. Is Russia experiencing a net capital inflow? 2. What is Russia's Total...
-
The following cost information was provided to you for analysis: September 12,000 Units Produced Costs: TIC TAC TOE TING August 10,000 P80,000 70.000 60.000 50,000 How much is the fixed cost per...
-
Does the reduction of acetoacetyl ACP in step 6 occur on the Re face or the Si face of themolecule? NADPH NADP+ SACP SACP H3C Acetoacetyl ACP B-Hydroxybutyryl ACP
-
Identify the two steps in glycolysis in which ATP is produced.
-
Look at the entire glycolysis pathway and make a list of the kinds of organic reactions that take place-nucleophilic acyl substitutions, aldol reactions, E1cB reactions, and so forth.
-
In which method does a company sequence the orders to do the work that is fastest to complete first? First-in, first-served Shortest process time Longest process time Earliest due date
-
Budgets must link to objectives, so budgeting is a tactical implementation of the Operations, business plan Strategies, operation plan Business plan, operation plan Strategies, business plan
-
The _ _ _ _ _ phase of the product life cycle sees the beginning of competition. The primary strategic goal during this period is to establish the product as firmly as possible in the marketplace....

Study smarter with the SolutionInn App


