Use the curve-fit coefficients for the standardized enthalpy from Table H.2 to verify the enthalpies of formation
Question:
Use the curve-fit coefficients for the standardized enthalpy from Table H.2 to verify the enthalpies of formation at 298.15 K in Table H.1 for methane, propane, and hexane.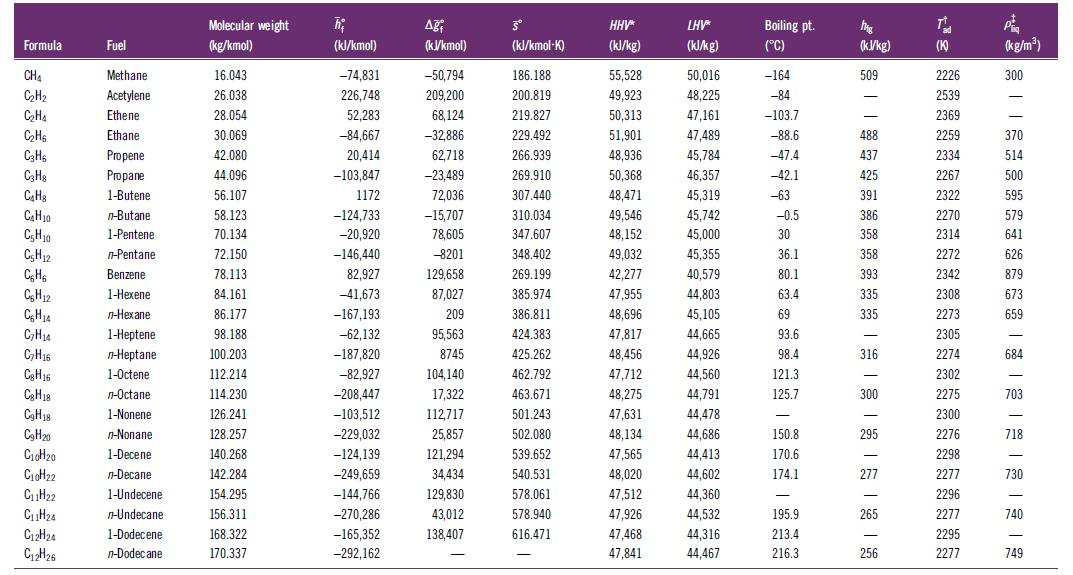
Transcribed Image Text:
Formula CHA C₂H₂ C₂H₁ C₂H6 C3H6 C3H8 C₂H C₂H₂0 C₂H10 C5H12 C₂H6 CH12 C₂H14 C₂H₂4 C₂H16 Cg H16 Cg H18 C₂H18 C₂H20 C₁0H₂0 C₁0H22 C₁1H₂2 C₁1H₂4 C12H24 C12H26 Fuel Methane Acetylene Ethene Ethane Propene Propane 1-Butene n-Butane 1-Pentene n-Pentane Benzene 1-Hexene n-Hexane 1-Heptene n-Heptane 1-Octene n-Octane 1-Nonene n-Nonane 1-Decene n-Decane 1-Undecene n-Undecane 1-Dodecene n-Dodecane Molecular weight (kg/kmol) 16.043 26.038 28.054 30.069 42.080 44.096 56.107 58.123 70.134 72.150 78.113 84.161 86.177 98.188 100.203 112.214 114.230 126.241 128.257 140.268 142.284 154.295 156.311 168.322 170.337 h₂ (kJ/kmol) -74,831 226,748 52,283 -84,667 20,414 -103,847 1172 -124,733 -20,920 -146,440 82,927 -41,673 -167,193 -62,132 -187,820 -82,927 -208,447 -103,512 -229,032 -124,139 -249,659 -144,766 -270,286 -165,352 -292,162 Aği (kJ/kmol) -50,794 209,200 68,124 -32,886 62,718 -23,489 72,036 -15,707 78,605 -8201 129,658 87,027 209 95,563 8745 104,140 17,322 112,717 25,857 121,294 34,434 129,830 43,012 138,407 - Šº (kJ/kmol-K) 186.188 200.819 219.827 229.492 266.939 269.910 307.440 310.034 347.607 348.402 269.199 385.974 386.811 424.383 425.262 462.792 463.671 501.243 502.080 539.652 540.531 578.061 578.940 616.471 - HHV* (kJ/kg) 55,528 49,923 50,313 51,901 48,936 50,368 48,471 49,546 48,152 49,032 42,277 47,955 48,696 47,817 48,456 47,712 48,275 47,631 48,134 47,565 48,020 47,512 47,926 47,468 47,841 LHV* (kJ/kg) 50,016 48,225 47,161 47,489 45,784 46,357 45,319 45,742 45,000 45,355 40,579 44,803 45,105 44,665 44,926 44,560 44,791 44,478 44,686 44,413 44,602 44,360 44,532 44,316 44,467 Boiling pt. (°C) -164 -84 -103.7 -88.6 -47.4 -42.1 -63 -0.5 30 36.1 80.1 63.4 69 93.6 98.4 121.3 125.7 - 150.8 170.6 174.1 - 195.9 213.4 216.3 MAR (kJ/kg) 509 - - 488 437 425 391 386 358 358 393 335 335 316 300 - 295 277 265 256 T (K) 2226 2539 2369 2259 2334 2267 2322 2270 2314 2272 2342 2308 2273 2305 2274 2302 2275 2300 2276 2298 2277 2296 2277 2295 2277 P (kg/m³) 300 370 514 500 595 579 641 626 879 673 659 - 684 - 703 - 718 730 740 749
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (9 reviews)
To verify the enthalpies of formation at 29815 K for methane propane and hex...View the full answer

Answered By

Sigei Kipngeno
I am a professional writer with more than 2 years of writing experience. I will always deliver a grade A paper with zero plagiarism . Thanks in advance.
4.70+
175+ Reviews
311+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
The standard free energies of formation and the standard enthalpies of formation at 298 K for difluoroacetylene (C2F2) and hexafluorobenzene (C6F6) are For the following reaction: C6F6(g) 3C2F2(g) a....
-
You are required to design an embedded system for a milk production line in a factory. A motor will control a conveyor belt that runs for 4 seconds to position five bottles under five nozzles. The...
-
The standard enthalpy of formation of H2O(l) at 298 K is 285.8 kJ/ mol. Calculate the change in internal energy for the following process at 298 K and 1 atm: H2O(l) H2(g) + O2(g) Eo = ?
-
Have you ever had to cope with the loss of a loved one? If so, what concepts described in this section provide context that may help you understand your experience and process of grieving?
-
Luby Corporation acquires a 100% business use automobile (MACRS 5-year recovery) on July 1, 2010 for $36,000. Luby does not elect Sec. 179, but the company otherwise desires to claim the maximum...
-
What attributes of a data code affect the likelihood of a recording error being made by a user of the code? Briefly outline some strategies to reduce error rates that occur with data codes.
-
What did Sherron Watkins say about rationalization and Jeff Skilling and Andy Fastow?
-
Assume that the auditors encountered the following sepa-rate situations when deciding on the report to issue for the current- year financial statements. 1. The auditors decided that sufficient...
-
Answer the following questions with explanation. 1) The most recent ?nancial statements for Crosby, |nc., follow. Sales for 2018 are projected to grow by 20 percent. Interest expense will remain...
-
Using the property data in Appendix D, reproduce Fig. 13.4. Note that the initial temperature and pressure are 298 K and 1 atm, respectively. Spreadsheet software is recommended to facilitate your...
-
Every Balance Sheet and Statement of Profit and Loss of a company shall comply with: A. US GAAP B. Indian Accounting Standard C. International Accounting Standard.
-
Which is worth more: (a) $500 today, (b) $1,000 in 9 years if the interest rate is 8 percent?
-
Nursing as a calling is changing as other thriving explanations behind living work with and close by clinical watchmen after a short time, and patients' information levels have improved. The measure...
-
Complete a two-page response to why law enforcement is struggling to recruit and retain officers in the U.S. Support your argument with sources (in-text citations and references). Also, provide...
-
Otto has the following strategy to save for his retirement. On January 1, 2022, he deposits $ 80,000 into a savings account. From then on, he will add an additional fixed amount x into his savings...
-
Design a slider-crank mechanism so that displacement of the slider is proportional to the crank rota- tion in the interval 30 0 100. Assume initial distance of the slider equal to 15 cm and final...
-
Q No.16 Abbas runs a business which retails high quality clothing. It is particularly busy during the festive season. The budgeted sales and purchases figures for September 2012 to January 2013 are...
-
Consider the following hypothetical data for 2012 and 2013: a. Find the price level for 2012 and 2013. What is the rate of inflation between the two years? b. What is the rate of inflation between...
-
Dr. Chan obtained a $15,000 demand loan at prime plus 1.5% on September 13 from the Bank of Montreal to purchase a new dental X-ray machine. Fixed payments of $700 will be deducted from the dentists...
-
Consider a simple ideal Rankine cycle and an ideal regenerative Rankine cycle with one open feedwater heater. The two cycles are very much alike, except the feedwater in the regenerative cycle is...
-
Reconsider Prob. 1034. How much does the thermal efficiency of the cycle change when the temperature at the entrance to the low-pressure turbine is increased to 550C? Data From Q#34: Consider a steam...
-
Consider a steam power plant that operates on the ideal reheat Rankine cycle. The plant maintains the boiler at 17.5 MPa, the reheater at 2 MPa, and the condenser at 50 kPa. The temperature is 550C...
-
Tides are considered a predictable source of energy because they rise and fall periodically: Group of answer choices due to the Coriolis effect due to the topography of the bottom of the ocean due...
-
Which statement below accurately states why most winds form? Multiple choice question. Air released by plants must displace the existing air around the plant, which in turn sets up a chain reaction...
-
(a) A circular cylindrical liquid jet of diameter, D, spontaneously breaks down into spherical droplets of diameter, d. Show that the minimum possible d/D = 1.5 due to surface tension/energy. (b)...

Study smarter with the SolutionInn App


