Potassium, an alkali metal, enters a condenser in the vapor phase with a mass flow rate of
Question:
Potassium, an alkali metal, enters a condenser in the vapor phase with a mass flow rate of 0.02 kg/s and at a temperature and pressure of 1370 K and 0.4 MPa. It exits as a saturated liquid at 0.2 MPa. Calculate the rate of energy transfer as heat from the condenser in kW using Appendix A.11 for data.
Data From A.11
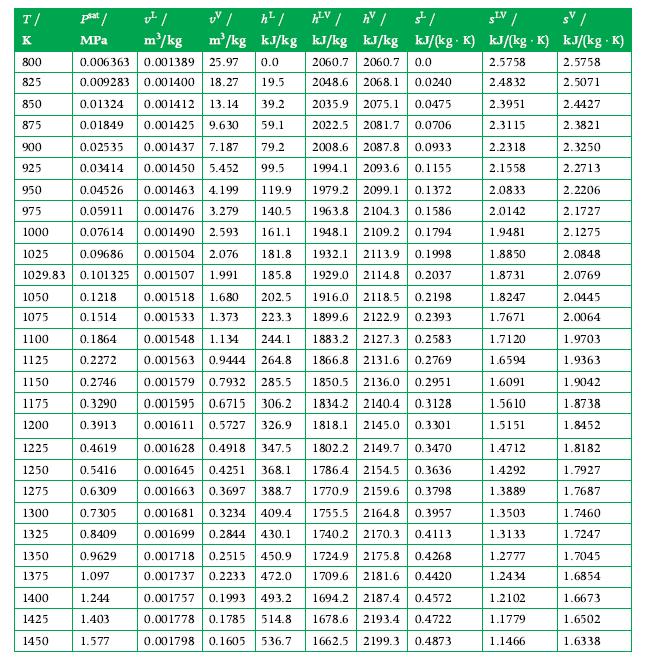
Transcribed Image Text:
T/ K 800 825 850 875 900 925 950 975 1000 1025 0.09686 1029.83 0.101325 0.1218 0.1514 0.1864 0.2272 0.006363 0.001389 25.97 0.0 0.009283 0.001400 18.27 19.5 0.01324 0.001412 13.14 39.2 2035.9 2075.1 0.0475 2022.5 2081.7 0.0706 0.02535 2008.6 2087.8 0.0933 0.01849 0.001425 9.630 59.1 0.001437 7.187 79.2 0.001450 5.452 99.5 1994.1 2093.6 0.1155 1979.2 2099.1 0.1372 1963.8 2104.3 0.1586 0.03414 0.04526 0.001463 4.199 119.9 0.05911 0.001476 3.279 140.5 161.1 0.07614 0.001490 2.593 0.001504 2.076 1948.1 2109.2 0.1794 1932.1 2113.9 0.1998 1929.0 2114.8 0.2037 0.001507 1.991 1050 0.001518 1.680 202.5 1916.0 2118.5 0.2198 1899.6 2122.9 0.2393 1075 0.001533 1.373 223.3 244.1 1100 0.001548 1.134 1883.2 2127.3 0.2583 1125 0.001563 0.9444 264.8 1866.8 2131.6 0.2769 1150 1175 1834.2 2140.4 0.3128 1818.1 2145.0 0.3301 1802.2 2149.7 0.3470 1786.4 2154.5 0.3636 1770.9 2159.6 0.3798 0.001579 0.7932 285.5 1850.5 2136.0 0.2951 0.001595 0.6715 306.2 1200 0.001611 0.5727 326.9 1225 0.4619 0.001628 0.4918 347.5 0.5416 0.001645 0.4251 368.1 0.6309 0.001663 0.3697 388.7 0.7305 0.001681 0.3234 409.4 0.8409 0.001699 0.2844 430.1 0.001718 0.2515 450.9 0.001737 0.2233 472.0 0.001757 0.1993 493.2 0.001778 0.1785 514.8 0.001798 0.1605 536.7 1755.5 2164.8 0.3957 0.9629 1.097 1740.2 2170.3 0.4113 1724.9 2175.8 0.4268 1709.6 2181.6 0.4420 1694.2 2187.4 0.4572 1678.6 2193.4 0.4722 1662.5 2199.3 0.4873 1.244 1.403 1.577 1250 1275 plat/ MPa 1300 1325 1350 1375 1400 1425 1450 v²/ h²/ m³/kg m³/kg kJ/kg 0.2746 0.3290 0.3913 HV/ hv/ है। SLV / sv / kJ/kg kJ/kg kJ/(kg. K) kJ/(kg K) kJ/(kg K) 2060.7 2060.7 0.0 2.5758 2.5758 2048.6 2.4832 2.5071 2.3951 2.4427 2.3115 2.3821 2.3250 2.2713 2.2206 2.1727 2.1275 2.0848 2.0769 181.8 185.8 2068.1 0.0240 2.2318 2.1558 2.0833 2.0142 1.9481 1.8850 1.8731 1.8247 1.7671 1.7120 1.6594 1.6091 1.5610 1.5151 1.4712 1.4292 1.3889 1.3503 1.3133 1.2777 1.2434 1.2102 1.1779 1.1466 2.0445 2.0064 1.9703 1.9363 1.9042 1.8738 1.8452 1.8182 1.7927 1.7687 1.7460 1.7247 1.7045 1.6854 1.6673 1.6502 1.6338
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
Mh 1 Neglecting kinetic energy at the inlet and outlet of the condenser the energy balance is Mh QMh ...View the full answer

Answered By

Krishnavendra Y
I am a self motivated financial professional knowledgeable in; preparation of financial reports, reconciling and managing accounts, maintaining cash flows, budgets, among other financial reports. I possess strong analytical skills with high attention to detail and accuracy. I am able to act quickly and effectively when dealing with challenging situations. I have the ability to form positive relationships with colleagues and I believe that team work is great key to performance. I always deliver quality, detailed, original (0% plagiarism), well-researched and critically analyzed papers.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics Fundamentals And Engineering Applications
ISBN: 9780521862738
1st Edition
Authors: William C. Reynolds, Piero Colonna
Question Posted:
Students also viewed these Engineering questions
-
The Stillwater geothermal power plant in Nevada, which started full commercial operation in 1986, is designed to operate with seven identical units. Each of these seven units consists of a pair of...
-
A power plant operates on a regenerative vapor power cycle with one closed feed water heater. Steam enters the first turbine stage at 10 MPa, 500oC and expands to 1 MPa, where some of the steam is...
-
HL Co. uses the high-low method to derive a total cost formula. Using a range of units produced from 1,500 to 7,500, and a range of total costs from $21,000 to $45,000, producing 2,000 units will...
-
Over the past 2 years, Jonas Cone has used a dollar-cost averaging formula to purchase $300 worth of FCI common stock each month. The price per share paid each month over the 2 years is given in the...
-
The following transactions took place at Grow-Right Garden Center during June 20X1. Grow-Right Garden Center uses a perpetual inventory system. Record the transactions in a general journal. Use 10 as...
-
Visit 10 Web sites that sell products or services and record the URL of each. Evaluate each site's published privacy policy in terms of the conditions need for compliance with the Safe Harbor...
-
In the GMP partnership (to which Elan seeks admittance), the capital balances of Mary, Gene, and Pat, who share income in the ratio of 6:3:1, are Mary ........ $240,000 Gene ......... 120,000 Pat...
-
1. ASSIGNMENT YOU ARE FREE TO CHOOSE WHICH COMPANY YOU WILL REPORT TO: - Supercell Company or - Finlayson Company. The length of the assignment in total is 5-7 pages. Return your reflection on Learn,...
-
Oxygen flows through an adiabatic steady flow compressor as saturated vapor at a rate of 1000 kg/hr. The saturated vapor enters at 2.5 bar and exits at 17.5 bar and 175 K. Calculate the shaft work...
-
An adiabatic pistoncylinder system contains a 1000 W immersion heater and 4 kg of water. The water is initially at 1 atm and 96% quality. The heater is operated for 7 minutes. Calculate the final...
-
Use the price data in exercise 18 to compute a Paasche index for the shipping cost if 2009 quantities are 4000, 3000, 7500, and 3000 for each of the four products. In exercise 18 Mean Shipping Cost...
-
The saturation pressure of liquid water at \(393.38 \mathrm{~K}\) is \(200 \mathrm{kPa}\). Estimate the fugacity of liquid water at \(372.12 \mathrm{~K}\) and \(300 \mathrm{kPa}\). We are given that...
-
Discuss the importance of generalized compressibility chart.
-
What is virial equation of state?
-
How do you explain the significance of virial coefficients?
-
Compare the Linde process with the Claude process for air liquefaction.
-
An executive recruitment firm used a lengthy online survey to gather information on the job mobility of mid-level managers. A sample of 500 eligible middle managers was selected, using a simple...
-
[a] Two foam blocks, each with a charge of 19 micro coulombs (1 C = 10-6 C), are both held in place 19 cm apart in the east-west direction. A foam ball with a charge 49 C is placed 55 cm north of the...
-
The Fischer esterification reaction is given by, Determine the equilibrium constant K of this reaction in terms of the concentrations of the reactants c R-(C=O)-OH , c R-OH and of the products c...
-
Steel wool is placed inside a cylinder filled with molecular oxygen O 2 , considered as an ideal gas. A piston ensures a constant pressure of the gas. The steel wool reacts with the molecular oxygen...
-
Apply the general definition of the battery potential, to the Daniell cell ( 8.7.4) and show that it yields relation (8.108). Show that the battery potential can be written as, where - 1 zFF A VaA PA
-
All costs in a homogeneous cost pool have Question 60 options: A) only one cost driver that will accurately represent the relationship between the costs and the activity. B) to be adjusted at year...
-
Which of the following is true for B-corps? Question 3 options: a) It is a for-profit corporation. b) It is a nonprofit corporation. c) They are taxed differently than c-corporations. d) They are...
-
Because interest earned on U.S. bonds and notes is not taxed by Oregon, special treatment is required if expenses related to this income is deducted as an itemized expense on your federal return....

Study smarter with the SolutionInn App


