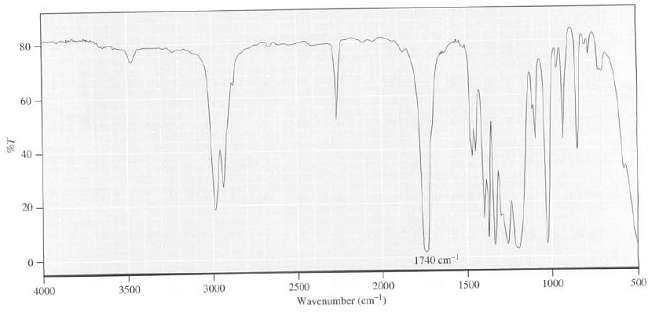
Suggest a possible structure for a compound with the formula C5H7O2 that has the following IR spectrum
Question:
Suggest a possible structure for a compound with the formula C5H7O2 that has the following IR spectrum and explain yourreasoning:
Transcribed Image Text:
80 60 40 20 - 1740 cm S0 1000 2000 1500 2500 3500 3000 4000 Wavenumber (em)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
The DU of this compound is 3 The peaks in the 30002...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Suggest a possible structure for a compound with the formula C5H12O that has the following IR spectrum and explain yourreasoning: 80 60 40 20 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm)
-
Suggest a possible structure for a compound with the formula C6H12O2 that has the following IR spectrum and explain your reasoning: 60 40 20 1741 cm 4000 3500 3000 2500 Wavenumber (em) 2000 1500 1000...
-
Suggest a possible structure for a compound with the formula C9H10O that has the following IR spectrum and explain yourreasoning: 100 - 50 1706 em- 4000 3500 3000 2500 2000 1000 500 1500 Wavenamber...
-
If you place a charged object next to an uncharged object thatis a conductor, If you place a charged object next to an uncharged object thatis a conductor, A) The force depends on whether the charge...
-
"On what basis shall we allocate resources to program A instead of program B," is the perennial statement in public sector budgeting. Identify and discuss at least five methodologies developed in the...
-
Successful ventures balance entrepreneurial characteristics with managerial style.
-
What book introduced the structural and behavioral consideration of fraud? What is the iceberg theory?
-
Given the rapid rate of change in both information technologies and business environments, do you believe that extensive information resource planning efforts are worthwhile for most organizations?...
-
You have been provided the following stock information on the Profitable Corporation: a) Calculate the return on this stock for each month. b) Calculate the arithmetic average return on the company...
-
Calculate the magnitude of smallest force F, such that resultant force has magnitude of 40 lb 20 Calculate the magnitude of smallest force F, such that resultant force has magnitude of 40 lb 20 lb F,...
-
Explain which functional group(s) is present in the compound that has this IRspectrum: 80 60 40 20 1721 cm 3500 4000 3000 2500 1500 2000 1000 S00 Wavenimber (cm)
-
Suggest a possible structure for a compound with the formula C7H12O that has the following IR spectrum and explain yourreasoning: 80 40 20 1718 cm 0- 500 1000 2000 1500 2500 3000 3500 4000 Wavenumber...
-
The correlation coefficient remains unchanged if we change the units used to measure x, y, or both. Determine whether the given property is true, and explain your answer.
-
Crimson Electronics, a company specializing in manufacturing smartphones, reports the following financials for the year ended 31 December 2022. Sales for the year: (8,000 units at $200 each) Direct...
-
Data for two variables, and y, follow. a. Develop the estimated regression equation for these data (to 2 decimals). Enter negative value as negative number. R 22 24 25 28 41 22 3 12 23 31 35 70 b....
-
The objective is to describe the major regulations that govern the conduct of financial institutions and corporations. The start of the new millennium saw several corporate scandals, the largest and...
-
Do the following program BOTH in pseudocode and submit that as pseudocode.txt , AND regular Java code that can run. Use methods when appropriate!! The program first asks the user for the password. If...
-
calculate work-in-process inventory: Purchased direct materials = $125,000 Direct Labor costs = 75,000 indirect labor costs = 25,000 other manufacturing overhead (excluding indirect labor) = 115,000...
-
When V-belt drive is preferred?
-
Pappa's Appliances uses the periodic inventory system. Details regarding the inventory of appliances at January 1, purchases invoices during the year, and the inventory count at December 31 are...
-
Suppose the propane-distribution industry currently fits all the characteristics of perfect competition. If the government were to decide to limit the number of propane distributors to a handful of...
-
What general kind of reaction does ATP carry out?
-
What general kind of reaction does NAD+ carry out?
-
What general kind of reaction does FAD carry out?
-
Do the winners from free trade win the political argument? Why or why not? The winners from free trade win the political argument because, OA. do, members of Parliament need their votes OB. do not it...
-
22 Statistics Canada has recently reported historically high increases in the CPI. As Tiff Macklin, the governor of the Bank of Canada, noted, higher inflation in Canada will help some Canadians, but...
-
Projectile Motion Component of Motion Characteristics I. Horizontal 1. Affected by gravity II. Vertical 2. Exhibits uniform motion 3. Exhibits uniform accelerated motion 4. Component of initial...

Study smarter with the SolutionInn App


