The 1H NMR spectrum shown is that of an ether with the formula C4H8O. Propose astructure. TMS
Question:
The 1H NMR spectrum shown is that of an ether with the formula C4H8O. Propose astructure.
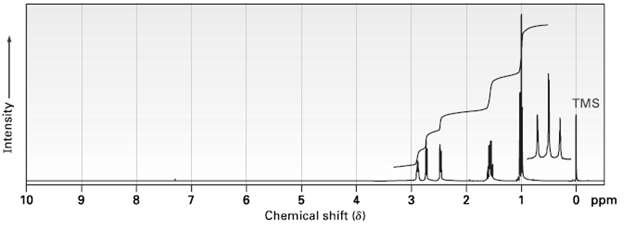
Transcribed Image Text:
TMS 10 9. 8. 7. 6. O ppm Chemical shift (8) Intensity 3.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (15 reviews)
CH3CHCCH H 12Epox...View the full answer

Answered By

Rohith Bellamkonda
I am studying in IIT Indore,the most prestigious institute of India.I love solving maths and enjoy coding
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The 1H NMR spectrum shown is that of 3-methyl-3-buten-l-ol. Assign all the observed resonance peaks to specific protons, and account for the splittingpatterns. TMS H H2H20 10 8. O ppm Chemical shift...
-
The 1 H NMR spectrum shown is that of a compound with formula C 9 H 10 O. How many double bonds and/or rings does this compound contain? If the unknown has an IR absorption at 1690 cm ?1 , what is a...
-
The 1 H NMR spectrum shown is that of a compound isomeric with the one in Problem 19.65. This isomer has an IR absorption at 1730 cm ?1 . Propose a structure. TMS 10 O ppm 1. Chemical shift (8)...
-
Provide a brief description of the seven basic steps to use JDBC.
-
Would you feel the same way about having biometric information on your driver's license as on your passport? Why or why not?
-
Write a paper describing in detail how the income statement, statement of owners equity, and balance sheet are related.
-
Is the Internal Revenue Code responsible for the backdating problem? Did Code Sec. 162(m), which restricts the deductibility of nonperformance-based executive compensation to \(\$ 1\) million, cause...
-
Stainless Refrigerator Company purchases ice makers and installs them in its products. The ice makers cost $138 per case, and each case contains 12 ice makers. The supplier recently gave advance...
-
Question 26 4 pts Company ABC is authorized to issue 10,000,000 shares of $2.50 par common stock by the state of Florida on 12/31/2008. On 1/1/2009, ABC issues 4,000,000 shares of common stock for...
-
The adjusted trial balance for Burlington Advertising Services is presented below: Requirements 1. Prepare the income statement for the year ending December 31, 2016. 2. Prepare the statement of...
-
2-Butene-1-thiol is one component of shunk spray. How would you synthesize this substance from methyl 2-butenoate? From 1,3-butadiene CH3CH=CHCOCH3 CH3CH=CHCH2SH 2-Butene-1-thiol Methyl 2-butenoate
-
Give IUPAC name for the following compounds (reddish brown =Br): (a) (b) (c)
-
In attributes sampling, why must error rates be estimated?
-
Robin Hood famously broke the rules so he could steal from the rich and give to the poor, because he felt the laws were unjustly applied to the common people. Robin Hood is operating at what level of...
-
What is your effective tax rate if you paid $27,621 in 2021 and your income was $140,000?
-
Identify a brand that utilizes marketing effectively. In your roundtable conversation, share how the product or service delivers customer value. To do so, think about the customers. What are their...
-
What ethical considerations arise in the context of globalization, particularly regarding issues of cultural appropriation, exploitation, and the responsibility of global actors toward vulnerable...
-
Beatrice needs $400,000 in her RRSP by the time she turns 65 to fully fund her retirement. If she is currently 25 years old and has nothing invested in her RRSP, how much money does she have to save...
-
Briefly explain what is permissible and what is prohibited under Rule 502-Advertising and Other Forms of Solicitation.
-
Write out the formula for the total costs of carrying and ordering inventory, and then use the formula to derive the EOQ model. Andria Mullins, financial manager of Webster Electronics, has been...
-
One kilogram of saturated liquid methane at 160 K is placed in an adiabatic pistion-and-cylinder device, and the piston will be moved slowly and reversibly until 25 percent of the liquid has...
-
Trans-1-Phenyl-1, 3-butadiene has max = 280 ( = 27,000) calculate the concentration of a solution that has A = 0.643 at 280nm in a 1 cm cell.
-
Nitro methane max = 275nm ( = 1.5) what kind of transition is responsible for this absorption?
-
3-Buten-2-one has max =213nm ( = 7080) and max = 320nm ( = 21) what kind of transition is responsible for each of these absorptions?
-
The company has its camera and drone assembly facilities in: a)Thailand b)South Korea c) the United States d)Japan e)Taiwan
-
Review the balance sheet for GHI Industries as of December 31, 2023, to assess its financial performance and stability. GHI Industries Balance Sheet As of December 31, 2023 Assets Current Assets Cash...
-
Question 8 What are the journal entries for recording the sale of an investment classified as held to maturity? (Assume the value of the investment has not changed since the original purchase, Debit...

Study smarter with the SolutionInn App


