The herbicide acifluorfen can be prepared by a route that begins with reaction between a phenol and
Question:
The herbicide acifluorfen can be prepared by a route that begins with reaction between a phenol and an aryl fluoride. Propose a mechanism.
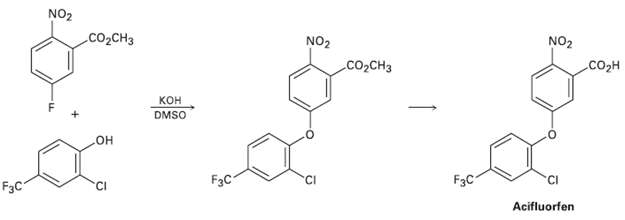
Transcribed Image Text:
NO2 CO2CH3 NO2 co,CH3 NO2 .CO2H кон DMSO HO F3C F3C CI FзC Acifluorfen
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (18 reviews)
The reaction is a nucleophilic aromatic subst...View the full answer

Answered By

Shailendra Tripathi
I am an Environmental Professional completed M.Sc. & Ph.D. in Environmental Science. Presented more than 50 research articles in International and National conference. Published more than 25 research paper in International and National scientific journals on waste management and water treatment. Written 2 books on Solid waste management and Vedic Environmental Science. Participated in the various training program on Wastewater analysis and sewage treatment. Proficiency includes sampling and laboratory analysis of wastewater and municipal solid waste. Hand on experience on the various laboratory protocol for environmental sample analysis. Experience on Data analysis software such as SPSS, Sigma stat etc. and Geographical modeling software such as Q-GIS, Diva GIS etc.
I am teaching B.Sc. and M.Sc. Biotechnology students in a State University affiliated college. The subject includes Environmental Biotechnology, Industrial Biotechnology, Fermentation Technology, Biostatistics, Research Methodology, Bioprocess Engineering.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
An carbonyl compound can be prepared by a reaction known as a selenenylation-oxidation reaction. A selenoxide is formed as an intermediate. Propose a mechanism for the reaction. 1. LDA/THF 2. CgHSeBr...
-
The herbicide oxyfluorfen can be prepared by reaction between a phenol and an aryl fluoride. Propose amechanism. FC. F3C. "CH-CH NO2 "CH-CH NO2 yfluorfen
-
An alkane with the formula C6H14 can be prepared by hydrogenation of either of only two precursor alkenes having the formula C6H12. Write the structure of this alkane, give its IUPAC name, and show...
-
A company has the following results for the three years to 31 October 2020: Assuming that all possible claims are made to relieve the trading loss against total profits, calculate the company's...
-
Why is continuous improvement, and programs such as CCC21 and Six Sigma, a good way to develop incremental innovation?
-
Following are 12 cost behaviour graphs. The total cost is shown on the y-axis and the volume (activity) is shown on the x-axis. For each of the following situations, identify the graph that most...
-
Gosh-bv- Golly Industries has adopted the follow ing production budget for the first 4 months of 2003. Each unit requires 5 pounds of raw materials costing $1.50 per pound. On December 31, 2002, the...
-
To what extent is the auditors opinion an indicator of a companys future financial success and future cash dividends to stockholders?
-
On January 1, Beckman, Inc., acquires 60 percent of the outstanding stock of Calvin for $45,780. Calvin Co. has one recorded asset, a specialized production machine with a book value of $13,400 and...
-
Suppose a $100,000 investment grows 3% during the first year and 4% during the second year. By what percent will it have grown after the two-year period?
-
Fluoxetine, a heavily prescribed antidepressant marketed under the name Prozac, can be prepared by a route that begins with reaction between a phenol and an alkyl chloride. (a) The rate of the...
-
The red fox (Vulpes) uses a chemical communication system based on scent marks in urine. Recent work has shown one component of fox urine to be a sulfide. Mass spectral analysis of the pure...
-
How may the effect of inflation be included in capital budgeting?
-
An aqueous solution of cobalt(II) iodide, CoI2, is made by dissolving 25.1 grams of cobalt(II) iodide in sufficient water in a 500. mL volumetric flask, and then adding enough water to fill the flask...
-
What is copiedNums' length after the code segment? int[] originalNums int[] copiedums = {0, 0, 0); copiedNums = copy (originalNums, 2); = {1, 2, 3, 4); public static int[] copy(int[] nums, int...
-
Volata Company began operations on January 1, 2023. In the second quarter of 2024, it adopted the FIFO method of inventory valuation. In the past, it used the LIFO method. The company's interim...
-
Find the graph of the equation. ???? = . 1 ???? 3 + . 2 ???? 2 - 4 ???? + 1
-
Amy and Tyler Smith are married, in their early forties, and have two 10 year old twins. Tyler works as an IT administrator and earns $70,000 each year. Amy is a project manager with a software...
-
Indicate the effects on the auditor's report when a change in an accounting principle is (a) made in conformity with GAAP (b) not made in conformity with GAAP.
-
Illini Company, Inc. Balance Sheet as of 12/31/20X0 Assets Current Assets: Cash $1,500,000 Accounts receivable, net 18,000 Inventory 50,000 Total current assets 1,568,000 Equipment 90,000 Goodwill...
-
The human body generates heat by the metabolism of carbohydrates and other food materials. Metabolism provides energy for all biological activities (e.g., muscle contractions). The metabolic...
-
Cetylethyldimethylammonium bromide is the common name for a compound with antiseptic properties. Predict its solubility behavior in water and in diethyl ether. Br
-
Write a three-dimensional formula for each of the following molecules using the wedge-dashed wedge-line formalism. If the molecule has a net dipole moment, indicate its direction with an arrow, . If...
-
Consider each of the following molecules in turn: (a) Dimethyl ether, (CH3)2O; (b) Trimethylamine, (CH3)3N; (c) Trimethylboron, (CH3)3B; and (d) Dimethylberyllium, (CH3)2Be. Describe the...
-
Rosie Dry Cleaning was started on January 1, Year 1. It experienced the following events during its first two years of operation: Events Affecting Year 1 Provided $34,850 of cleaning services on...
-
The IMA's Statement of Ethical Professional Practice includes Standards of conduct for Management Accountants. The standard that includes management accountants responsibility to provide all relevant...
-
At the beginning of the period, the Cutting Department budgeted direct labor of $63,920 and supervisor salaries of $47,190 for 3,760 hours of production. The department actually completed 4,100 hours...

Study smarter with the SolutionInn App


