The metal ion indicator xylenol orange (Table 11-3) is yellow at pH 6 ( max = 439
Question:
The metal ion indicator xylenol orange (Table 11-3) is yellow at pH 6 (λmax = 439 nm). The spectral changes that occur as VO2+ is added to the indicator at pH 6 are shown here. The mole ratio VO2+/xylenol orange at each point is
-1.png)
Suggest a sequence of chemical reactions to explain the spectral changes, especially the isosbestic points at 457 and 528 nm.
-2.png)
Table 11-3
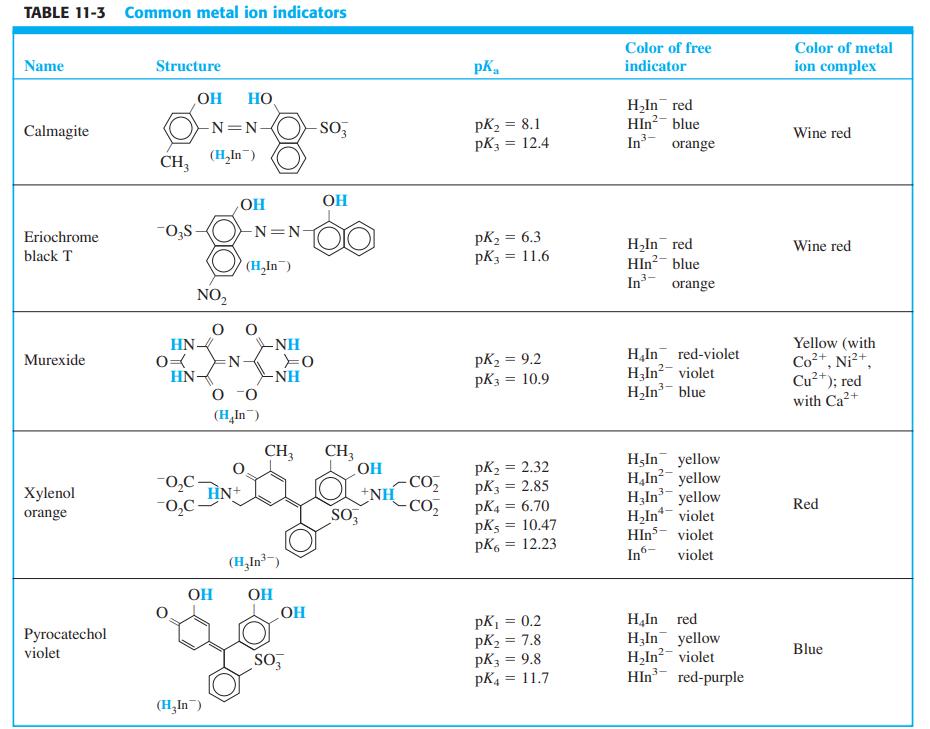
Transcribed Image Text:
Trace Mole ratiTrace Mole ratio Trace Mole ratio 0.60 0.70 0.80 0.90 1.0 12 13 1.5 14 2.0 15 3.1 16 4.1 1.3 0.10 2 0.20 0.30 0.40 5 0.50 4 10 16 0.1 Absorbance unit 13 12 600 500 Wavelength (nm) 400
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (11 reviews)
As VO 2 is added traces 19 the peak at 439 decreases a...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
A 1.000-mL sample of unknown containing Co2+ and Ni2+ was treated with 25.00 mL of 0.03872 M EDTA. Back titration with 0.02127 M Zn2+ at pH 5 required 23.54 mL to reach the xylenol orange end point....
-
A novel metal ion adsorbent material based on the bio polymer poly glucosamine is cast into a gel bead. The amine groups on the bio polymer have a high affinity for transition metal ions at...
-
The amount of lactic acid, HC3H5O3, produced in a sample of muscle tissue was analyzed by reaction with hydroxide ion. Hydroxide ion was produced in the sample mixture by electrolysis. The cathode...
-
Does the fact that the strategic petroleum reserve has never been used to offset shortfalls caused by an embargo mean that the money spent in creating the reserve has been wasted? Why or why not?
-
Farwell Company closes its books monthly. On September 30, selected ledger account balances are: Notes Receivable .......$37,000 Interest Receivable ....... 183 Notes Receivable include the...
-
An aluminum transmission line with a diameter of 20 mm has an electrical resistance of R' elec = 2.636 x 10 -4 n/m and carries a current of 700 A. The line is subjected to frequent and severe cross...
-
Use the comparative statements from Application Problem 17-1 to complete this problem. Instructions: 1. Based on CyberOptic's comparative financial statements prepared in Application Problem 17-1 and...
-
On March 3, Pitrof Appliances sells $710,000 of its receivables to American Factors Inc. American Factors Inc. assesses a service charge of 4% of the amount of receivables sold. Instructions Prepare...
-
On January 1 , 2 0 1 4 , Tong purchased a ten - year bond bought at par. The bond pays semiannual coupons. Because Tong is an actuary, he enjoys calculating duration as a hobby. Tong calculated the...
-
A bottle-filling line uses a feeder screw mechanism, as shown in Figure AP6.4. The tachometer feedback is used to maintain accurate speed control. Determine and plot the range of K and p that permits...
-
When are isosbestic points observed and why?
-
Infrared spectra are customarily recorded on a transmittance scale so that weak and strong bands can be displayed on the same scale. The region near 2 000 cm -1 in the infrared spectra of compounds A...
-
With regard to the selection process, what is meant by the term organizational fit?
-
1. For the structure shown in Figure 1, determine (i) the unknown displacements at all the considered nodes, (ii) the strains as well as (iii) the stresses for each and every of the involved constant...
-
1. Amsterdam Company owns the mineral rights to land that has a deposit of ore. The company in uncertain if it should purchase equipment and open a mine on the property. After careful study, the...
-
A company acquired as a long - term investment $ 1 5 0 million of 4 . 0 % bonds, dated July 1 , on July 1 , Year 1 . Company management has the positive intent and ability to hold the bonds until...
-
1. What cultural aspects are likely to affect the negotiation? 2. To what extent will gender play a role in this negotiation? 3. What are your two main objectives in the negotiation withMr Hagashi?...
-
Abby is single and age 25. She has AGI of $110,000 and incurs the following expenditures in 2022: Medical expenses: $9,000 Interest on home mortgage (mortgage was $300,000): $11,000 State income tax:...
-
Condensed statement of financial position and comprehensive income statement data for Clack Ltd. follow: Additional information: 1. The allowance for doubtful accounts was $4,000 in 2014 and $5,000...
-
Consider a closed, rigid tank with a volume of 0.8L, filled with cold water initially at 27C. The tank is filled such that there are no voids (air pockets) within. The initial pressure within the...
-
Select indicators from Table 10-3 that would be useful for the titrations in Figures 10-1 and 10-2 and the pK a = 8 curve in Figure 10-3. Select a different indicator for each titration and state...
-
When 100.0 mL of a weak acid were titrated with 0.093 81 M NaOH, 27.63 mL were required to reach the equivalence point. The pH at the equivalence point was 10.99. What was the pH when only 19.47 mL...
-
A 0.100 M solution of the weak acid HA was titrated with 0.100 M NaOH. The pH measured when V b = V e was 4.62. Using activity coefficients, calculate pKa. The size of the A - anion is 450 pm.
-
You are evaluating a closed-end mutual fund and see that its price is different from its net asset value (NAV). The fund has an expense ratio (s) of 2.80% and a dividend yield (5) of 4.00%. The fund...
-
Here is your question to solve, due April 8 . A $ 1 4 , 0 0 0 bond has 8 years until maturity with a semi - annual coupon rate of 5 . 8 % . Determine its market price at a semi - annual market rate...
-
Green Moose Industries is a company that produces iGadgets, among several other products. Suppose that Green Moose Industries considers replacing its old machine used to make iGadgets with a more...

Study smarter with the SolutionInn App


