Using the y-x and T-y-x diagrams in Figures 4.3 and 4.4, determine the temperature, amounts, and compositions
Question:
Using the y-x and T-y-x diagrams in Figures 4.3 and 4.4, determine the temperature, amounts, and compositions of the equilibrium vapor and liquid phases at 101 kPa for the following conditions with a 100-kmol mixture of nC6 (H) and nC8 (C).(a) ZH = 0.5, ? = V/F = 0.2(b) ZH = 0.4, yH = 0.6(c) ZH = 0.6, xc = 0.7(d) ZH = 0.5, ? = 0(e) ZH = 0.5, ? = 1.0(f) ZH = 0.5, T = 200?F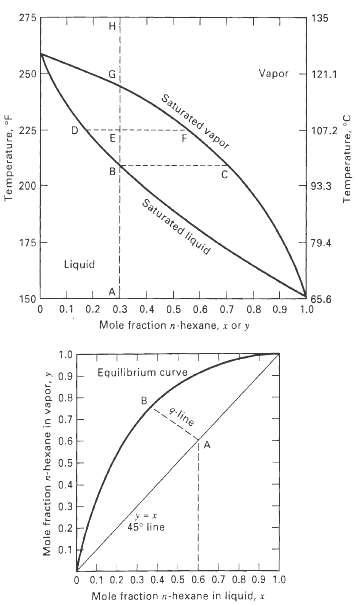
Transcribed Image Text:
135 275 121.1 Vapor 250 Saturated vapor 107.2 225 B. 93.3 200 Saturated liquid 79.4 175 Liquid 65.6 1.0 0.8 0.9 150 0.2 0.3 0.4 0.5 0.6 0.7 0.1 n hexane, x or y Mole fraction 1.0 Equilibrium curve 0.9 0.8 q-line 0.7- 0.6 0.5 0.4- 0.3 45° line 0.2 0.1 0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1.0 Mole fraction n-hexane in liquid, x Temperature, °F Mole fraction n-hexane in vapor, y Temperature, °C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
Let z H mole fraction of nhexane in the feed and VF Use inverse leverarm ...View the full answer

Answered By

Shadrack Mulunga
I am a Biochemistry by profession. However, I have explored different fields of study. My quest to explore new fields has helped me gain new knowledge and skills in Business, clinical psychology, sociology, organizational behavior and general management, and Project Management. I count my expertise in Project management, in particular, creation of Work Break Down Structure (WBS) and use of Microsoft Project software as one of my greatest achievement in Freelancing industry. I have helped thousands of BSC and MSC students to complete their projects on time and cost-effectively using the MS Project tool. Generally, I find happiness in translating my knowledge and expertise to success of my clients. So far, i have helped thousands of students to not only complete their projects in time but also receive high grades in their respective courses. Quality and timely delivery are the two key aspects that define my work. All those who hired my services always come back for my service. If you hire my services today, you will surely return for more. Try me today!
5.00+
154+ Reviews
289+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Q1. Suppose player A and player B are playing the following game. Player A can choose "Up" and "Down", while player B can choose "Left" and "Right". The payoff matrix is depicted below. Player A's...
-
Liquid water at 200 kPa and 20C is heated in a chamber by mixing it with superheated steam at 200 kPa and 300C. Liquid water enters the mixing chamber at a rate of 2.5 kg/s, and the chamber is...
-
A mixture of 1 kmol carbon dioxide, 2 kmol carbon monoxide, and 2 kmol oxygen, at 25C, 150 kPa, is heated in a constant pressure SSSF process to 3000 K. Assuming that only these same...
-
Describe six danger signs of a bust-out artist.
-
The following ratios are available for Tianjin Inc.: Instructions (a) Is Tianjin's liquidity improving or worsening? Explain. (b) Do changes in turnover ratios affect profitability? Explain. (c) Do...
-
Write several clear, measurable objectives for the training to be delivered to the employees in the customer order group.
-
11. Harper & Row signed a contract with former President Gerald Ford to publish his memoirs. As part of the deal, the two agreed that Time magazine could print an excerpt from the memoirs shortly...
-
Rivera Company has several processing departments. Costs charged to the Assembly Department for November 2012 totaled $2,280,000 as follows. Production records show that 35,000 units were in...
-
Please help!!! Due tonight and I suck at these types ofquestions ???? THANK YOU Excel Online Structured Activity: Personal taxes Mary Jarvis is a single individual who is working on filing her tax...
-
In 2018, your company purchased a front end loader for $150,000, and a dump truck for $85,000. The front end loader was placed in service in April, 2018 and the dump truck was placed in service in...
-
Vapor-liquid equilibrium data for mixtures of water and isopropanol at 1 atm (101.3 kPa, 760 torr) are given below. (a) Prepare T-x-y and x-y diagrams. (b) When a solution containing 40 mol %...
-
For a binary mixture of components 1 and 2, show that the equilibrium phase compositions and amounts can be computed directly from the following reduced forms of Eqs. (5), (6), and (3) of Table 4.4 X...
-
What are the magnitude and direction of the current in the circuit in Figure P31.45? Data from Figure P31.45 18.0 V 10 ww 9.0 V
-
Given an = 3- -n 1. n and bn = -7. ( })" determine whether (an + b) is convergent n=0 or divergent and determine its value. Enter a real number (the value of the series) if the series is convergent,...
-
Summarize this paragraph to fit into 2-3 slides for a power-point presentation. Only take out important key elements. Two potential pricing strategy options for a B2B market could be cost-plus...
-
Derek Wilson operates Clean Ride Enterprises, an auto detailing company with 20 employees. Jamal Jackson has recently been hired by Wilson as a controller. Clean Ride's previous accountant had done...
-
Fill in the missing data for each of the following independent cases. (Ignore income taxes.) Note: Do not round intermediate calculations. Leave no cells blank - be certain to enter "0" wherever...
-
Sarahs Delights is a bakery in Stellenbos and employs 18 people, including: The manager of the savoury section is senior baker, Mandie who oversees 5 junior bakers. The manager of the sweet section...
-
Calculate 2 and 2 for the Maternal Adaptation data in Exercise 7.19. Exercise 7.19 Nurcombe, Howell, Rauh, Teti, Ruoff, and Brennan (1984) conducted an intervention program with mothers of low...
-
Use the information given about the angles and to find the exact value of: (a) sin( + ) (b) cos( + ) (c) sin( - ) (d) tan ( + ) (e) sin(2) (f) cos (2) (g) sin /2 (h) cos/2 cos = 4/5, 0 < < /2; cos =...
-
Discuss the ethical standards of transparency, trust, and social responsibility for mutual funds.
-
Compare the required solvent volume, stage number, and purity obtained when penicillin F is extracted from water preferentially to penicillin K into amyl acetate at pH 3.1 relative to the values at...
-
For each of the distillations D-1 and D-2 indicated in Figure 9.20, establish the type of condenser and an operating pressure.
-
A monoacidic sugar extracted from water into hexanol has distribution coefficients of 6.0 and 0.47 moles per liter at pH 4.0 and 5.5, respectively. Estimate the value of K D at pH 7.2.
-
D Question 10 4 pts Individuals may use reasonable force to protect individuals, themselves, and their property. Under the majority rule, reasonable force means: O deadly force is reasonable if an...
-
Liability Laws Select each description to either Common Negligence Laws or Dram Shop Liability Laws to indicate the type of law it describes. Apply only to licensees and their employees Based on...
-
Disney Parks, Experiences and Products, Inc. has positioned itself as offering happiness, magic, enchantment, and fun. Taglines for Disney's theme parks have included The Happiest Place on Earth,...

Study smarter with the SolutionInn App


