What alcohols would give the following products on oxidation? (b) (c) C (a) C
Question:
What alcohols would give the following products on oxidation?
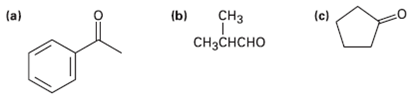
Transcribed Image Text:
(b) (c) Cнз (a) Cнзснсно
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (18 reviews)
Strategy Aldehydes are synthesized from oxid...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What starting material would give the following compound in an aliphatic Claisen rearrangement? CH,-CH-C
-
Give structures of the alkenes that would give the following products upon ozonolysis-reduction. (a) (b) and CH,CH,CH,_C_H cyclohexanone CH,-CH-_-_CH
-
The enzymatic oxidation of alkanes to produce alcohols is a simplified version of the reactions that produce the adrenocortical steroid hormones. In the biosynthesis of corticosterone from...
-
On 13 May 2014, Ruben acquired 400 shares in Xantan Ltd at a cost of 1,800. On 17 July 2020, the company made a 1 for 8 bonus issue and (on the same day) Ruben sold his bonus shares for 7 each....
-
What does "patching" refer to? Describe and illustrate two rules that might guide managers to build value in their businesses.
-
An all-equity firm has 100,000 shares outstanding worth $10 each. The firm is considering a project requiring an investment of $400,000 and has an NPV of $50,000. The company is also considering...
-
Give some examples of external fraud.
-
J. Ackbar, a former professional tennis star, operates Ackbar's Tennis Shop at the Miller Lake Resort. At the beginning of the current season, the ledger of Ackbar's Tennis Shop showed Cash $2,200,...
-
The difference between the cost of an equipment and its related accumulated depreciation is referred to as the: O a. depreciated difference of the equipment O b. fair value of the equipment O c. book...
-
As the HR Director of a U.S.-based company that is looking at global opportunities in China, you have been asked by the company president to prepare an outline for an HR strategic plan as part of the...
-
What product(s) would you expect from dehydration of the following alcohols with POCl3 in pyridine? Indicate the major product in eachcase? (a) (b) (c) CH3CH2CHCHCH3 CH CH3 H (d) (e) CCCH2CH...
-
What products would you expect from oxidation of the following compounds with CrO3 in aqueous acid with pyridinium chlorochromate? (a) 1-Hexanol (b) 2-Hexanol (c) Hexanol
-
Solve the following proportions for the unknown quantities 88:17 = a:45
-
In a program, a dictionary contains lists of students and their courses. The teacher is interested to have a dictionary that has the courses as key and the students enrolled in each course as values....
-
Show that the commutator [1,6] = -2, where =2-d and =+a. We will show that is the lowering operator for the harmonic oscillator wavefunctions.
-
"Maintainability is par excellence, a yardstick by which the quality of software can be judged. Paradoxical as it may seem, software that has been developed with a view to being changed is likely to...
-
Lone Star Meat Packers is a major processor of beef and other meat products. The company has a large amount of T - bone steak on hand and is deciding whether to sell the T - bone steaks as they are...
-
Steve Reese is a well-known interior designer in Fort Worth, Texas. He wants to start his own business and convinces Rob O'Donnell, a local merchant, to contribute the capital to form a partnership....
-
Briefly describe the SEC's rules concerning (a) qualifications of independent accountants and (b) accourtant's reports.
-
The words without recourse on an indorsement means the indorser is: a. not liable for any problems associated with the instrument. b. not liable if the instrument is dishonored. c. liable personally...
-
Of the following liquids at 20 C, which has the smallest viscosity? (a) Dodecane, C 12 H 26 ; (b) n-nonane, C 9 H 20 ; (c) n-heptane, C 7 H 16 ; (d) n-pentane, C 5 H 12 .
-
Using the pKa values of analogous compounds in Table 3.1, predict which would be the stronger base. (a) (b) (c) (d) or (CHa),Cor O: HO or
-
The pKa of the anilinium ion (C6H5NH3) is 4.63. On the basis of this fact, decide whether aniline (C6H5NH2) is a stronger or weaker base than methylamine.
-
Predict the outcome of the following reaction. -NH2
-
Multiple Choice Question Select the appropriate verb to complete the sentence. Pour rparer une machine, vous avez besoin de/d' _Blank_______ de bons outils (good tools). Multiple choice question....
-
In order for journalism to get high density, nutritious information to citizens, it takes a lot of... Group of answer choices consolidation money both of the answers listed
-
Select all that apply On 2/1, Sophie Company sold merchandise on account to Lexie Company in the amount of $1,500 and credit terms of 1/10, n/30. On 2/3, Lexie returned defective merchandise in the...

Study smarter with the SolutionInn App


