What alkyl halides would you use to prepare the following ketones by an acetoacetic ester synthesis? CH
Question:
What alkyl halides would you use to prepare the following ketones by an acetoacetic ester synthesis?
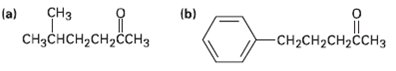
Transcribed Image Text:
CHз (a) (b) CHзснсH,сH,H3 -CH-CH2CH2CCHЗ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
As in the malonic ester synthesis you should identify the stru...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What alkyl halides would you use to prepare the following -amine acids by the amidomalonate method? (a) Leucine (b) Histidine (c) Tryptophan (d) Methionine
-
What reagents would you use to prepare the following compounds? a. b. CH CCH CH:C 0 CH3CCH-CH-CH (COCH-CH3 )2
-
What alkyl halides would you utilize to synthesize the following compounds, using the organoborane shown? a. b. c.
-
What is the value of a 13% coupon bond that is otherwise identical to the bond described in Part D? Would we now have a discount or a premium bond?
-
List the guidelines for effective headings.
-
Sandburg and Williams are the owners of a partnership that manufactures commercial lighting fixtures. Profits are allocated among the partners as follows: Sandburg was divorced as of the beginning of...
-
Assume the same information in Exercise 17-57 except that the lessee also paid legal fees in the execution of the lease of \(\$ 1,800\) on January \(1,2020\). Required a. How would Abel Company...
-
Depreciation?SYD, Act., SL, and DDB the following data relate to the Plant Assets account of Eshkol, Inc. at December 31, 2010. *In the year an asset is purchased, Eshkol, Inc. does not record any...
-
A manufactured product has the following information for June. Direct materials Direct labor Overhead Units manufactured Standard Quantity and Cost 7 pounds $8 per pound 3 DLH @ $16 per DLH 3 DLH @...
-
You are part of a research team investigating the effects of aerobic cardiovascular and moderate weight resistance training on the BMI (Body Mass Index) of moderately overweight but not obese teens...
-
How could you use a malonic ester synthesis to prepare the following compound?
-
Which of the following compounds cannot be prepared by an acetoacetic ester synthesis? Explain. (a) Phenyl acetone (b) Acetophenone (c) 3, 3-Dimethyl-2-butanone
-
Ira Koslowsky, a star employee, was on the fast track at Grandiose Private Equity, Inc. On his own he had borrowed money, creating a stake for himself of about $1 million in LUBICATe, an...
-
Silven Industries, which manufactures and sells summer lotions and insect repellents, has decided to diversify in order to stabilize sales throughout the year. A natural area for the company to...
-
Cape Town Footwear ( Pty ) Ltd ( \" Cape Town Footwear\" oncompany\" ) is a companyesident in South Africa. It specializesmanufacture of footwear andoperates from afacility located in Cape Town. The...
-
a. Discuss at least 5 reasons why the Product Owner should be a single person and not a committee? b. How can the Product Owner maintain authority over the product while collaborating with the Dev...
-
Recorded a $ 4 , 5 0 0 purchase of merchandise that actually applies to next year. Accrued $ 2 5 0 of interest revenue that actually applies to next year. Failed to accrue wages of $ 2 , 0 0 0 ....
-
The solution to dy = 2 e**y with initial condition (0, -In 5) can be expressed as y = -In (j - ke) for real numbers j dx and k. What are the values of j and k?
-
4-41 An external auditor is interested in whether a company has implemented an ef- fective ERM because: a. It reduces the likelihood that an organization will fail. b. It provides a framework for the...
-
How can you tell from the vertex form y = a(x - h) 2 + k whether a quadratic function has no real zeros?
-
What is hybridization? Why is hybridization necessary in valence bond theory?
-
Rank the following compounds in order of increasing reactivity (least reactive first) in an SN1 solvolysis reaction in aqueous acetone. Explain your answers. (The structure of tert-cumyl chloride is...
-
Explain why compound A reacts faster than compound B when they undergo solvolysis in aqueous acetone. CH C-Cl CH3 CH CH
-
A hydrocarbon A, C9H12, is treated with A-bromosuccinimide in CCl4 in the presence of peroxides to give a compound B, C9H11Br. Compound B undergoes rapid solvolysis in aqueous acetone to give an...
-
Shott Farm Supplies Corporation purchased 950 shares of General Motors stock at $53 per share and paid a brokerage fee of $1,500. Two months later, the shares were sold for $56 per share. The...
-
Anderton Corporation has provided the following production andaverage cost data for two levels of monthly production volume. Thecompany produces a single product. Production volume Directmaterials 2...
-
Sound Tek Inc. manufactures electronic stereo equipment. Themanufacturing process includes printed circuit (PC) board assembly,final assembly, testing, and shipping. In the PC board assemblyoSound 2...

Study smarter with the SolutionInn App


