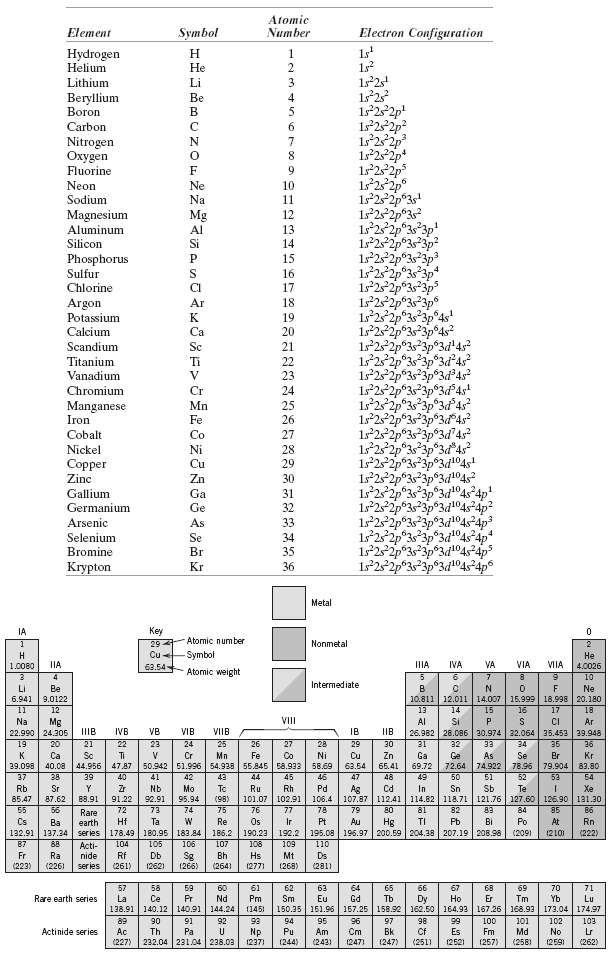
Without consulting figure?or Table 2.2, determine whether each of the electron configurations given below is an inert
Question:
Without consulting figure?or Table 2.2, determine whether each of the electron configurations given below is an inert gas, a halogen, an alkali metal, an alkaline earth metal, or a transition metal. Justify your choices.
(a) 1s22s22p63s23p63d74s2
(b) 1s22s22p63s23p6
(c) 1s22s22p5
(d) 1s22s22p63s2
(e) 1s22s22p63s23p63d24s2
(f) 1s22s22p63s23p64s1
Transcribed Image Text:
Atomic Number Electron Configuration Element Symbol 1s' 12 Hydrogen Helium Н Не 2 Lithium Li 122 1s°2s2p! 1s 22p? 1s 22p 1s 2-2p 12 2p 122p° 1s°2s2p°3s 1s°25 2p°3 Is22p°3s 3p 1s 2 2p° 1s 25 2p Beryllium Boron Be 4 Carbon 6. Nitrogen Охудen Fluorine Neon Sodium Ne 10 Na 11 Mg Magnesium Aluminum 12 Al 13 14 Silicon Phosphorus Sulfur 15 16 1s22p 3p° Chlorine 17 122 Argon Potassium Calcium Ar 18 19 1s 2 2p 1s 252 l2s 1s°2 1s2 ILs22 1s°2 2p 1s°2s 2p 1 2s 2p 1s 2s Is°2s Ca 20 Scandium Sc 21 Titanium 22 Vanadium 23 Chromium Cr 24 Manganese Iron Mn 25 Fe 26 Cobalt Co 27 Nickel Ni 28 Copper Zinc Gallium Cu 29 Zn 30 Ga 31 1s°25 2 1s 25 Germanium Ge 32 Arsenic Selenium As 33 Se 34 1525 1s°22 Bromine Br 35 s-4p° Krypton Kr 36 Metal Key 29 Cu - 63.54 IA Atomic number Nonmetal Symbol H. 1.0080 Не TIA IIIA IVA VA VIA VIIA 4.0026 Atomic weight 10 Intermediate Li Be Ne 10.811 12.011 14.007 15.999 18.998 20.180 15 6.941 9.0122 11 12 13 14 16 17 18 Na Mg AI Si P. Ar IIIB IVB VB VIB VIIB IB IB 22.990 24.305 26.982 28.086 30.974 32.064 35.453 39.948 23 19 20 21 22 24 25 26 27 28 29 30 31 32 33 34 35 26 Ca 40.08 So Ti 44.956 47.87 Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 50.942 51.996 | 54.938 | 55.845 58.933 72.64 74.922 78.96 79.904 39.098 58.69 63,54 65.4L 69.72 83.80 49 37 38 39 40 41 42 43 44 45 46 47 48 50 51 52 52 54 Rb Sr Nb Mo Te Ru Rh Pd Ag Cd In Sn Sb Te Xe 101.07 102.91 106.4 107.87 112.41| 114.82 | 118.71 | 121.76 | 127.60 126.90 131.30 76 85.47 87.62 88.91 91.22 92.91 95.94 (98) 56 77 79 80 86 55 72 73 74 75 78 81 82 83 84 85 Rare earth Cs Hf Ta Re Os Ir Pt Au Hg TI Pb Bi Po At Rn Ba 178.49| 180.95 183,84 186.2 190.23 132.91 137.24 192.2 195.08 196.97 200.59 204.38 207.19| 208.98 (2091 12100 (222) series Acti- nide 87 88 104 105 106 107 108 109 110 Fr Ra Rf Db Sg Bh Hs Mt Ds (223) (226) (261) 1262) (266) (2641 12771 (268) (2811 ser les 57 58 59 60 61 62 63 64 65 66 67 68 69 70 71 Rare earth series La Ce Pr Nd Pm Dy 150.35 | 161.96 167.25 158.92 162.50 | 164.93 167.26 | 168.93 173.04 | 174.97 Sm Eu Gd Tb Ho Er Tm Yb Lu 138.91 | 140.12 140.91 | 144.24 (145 89 90 91 92 94 95 96 97 98 99 100 101 102 103 Lr Actinide series Ac Np 232.04 231.04 | 238.03 Th Pa Pu Am Cm Bk Cf Es Fm Md No (227) 1237) (244) (243) 12471 (247) (2511 12521 (257) (2581 (2591 (262)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
a Electron configuration is that of a transition meta...View the full answer

Answered By

Rinki Devi
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions.
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students.
I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and helped them achieve great subject knowledge.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
Determine whether each of the following represents loss aversion. a. Nearing retirement, an investor chooses investments with lower return and lower risk, because she wants to make sure she has a...
-
Determine whether each of the functions 2n+1 and 22n is O(2n).
-
Determine whether each of the following scenarios best reflects features of Sweezy, Cournot, Stackelberg, or Bertrand duopoly: a. Neither manager expects her own output decision to impact the other...
-
Determine the equations for the voltage and instantaneous Power in the network in Figure P 9.2 4 2225 A Figure P 92
-
How does the weight of mercury in a barometer compare with the weight of an equal cross-section of air from sea level to the top of the atmosphere?
-
What do you think Campbell should do to counter the competition and remain in the top of the soup business?
-
Near the end of 2017, Royalty Corporation drilled an exploratory well that found oil, but not in commercially producible quantities unless the price of oil went up from $100 per barrel to $150 per...
-
Calculate the 90% confidence interval for the following variables, comparing lower, working, middle, and upper classes (CLASS) in the GSS sample. First, tell SPSS that we want to select all cases in...
-
1. An equity investment in less than 20% of another companys outstanding stock is accounted for using the fair value method. True False 2. Available-for-sale securities are securities that management...
-
Refer to Fig. 4.40. 8,0 in Semicircular hatch 10 in 26 in 20-in radius Ethylene glycol sg = 1.10 10 in Tank is symmetrical 30
-
To what group in the periodic table would an element with atomic number 114 belong?
-
(a) What electron subshell is being filled for the rare earth series of elements on the periodic table? (b) What electron subshell is being filled for the actinide series?
-
Neutron stars are composed of solid nuclear matter, primarily neutrons. Assume the radius of a neutron is approximately 1.0 * 10 -13 cm. Calculate the density of a neutron. [For a sphere V = (4/3)r 3...
-
Use the following information to answer the following question: Month Meals Served Utilities Costs December 45 401.00 January 35 360.00 February 25 347.00 March 40 385.00 April 50 414.00 20 Using the...
-
How might you assess the benefits of a training program designed to teach employees how to use the Internet to monitor stock prices? Use the Kirkpatrick model presented in book and in the video...
-
Should you be covered with life insurance? Why or Why not How will you best plan for life insurance needs?
-
Do you believe that the tourism industry should participate in a free-market system, or do you believe in market interventions? If so, what interventions? If not, why not? Defend your position.
-
Brandon Consulting Company is headquartered in Atlanta and has branch offices in Nashville and Birmingham. Brandon uses an activity-based costing system. The Atlanta office has its costs for...
-
A survey of 214 of the seniors graduating with a bachelor of science degree from a university found that 15% planned to obtain entry-level jobs in the health field. In Exercises 2534, identify the...
-
What are the main distinctions between the different schools of legal interpretation?
-
Compute or approximate the corresponding function values and derivative values for the given area function. In some cases, approximations will need to be done via a Riemann sum. = S T(x) = tan 0 de....
-
Rhenium has an HCP crystal structure, an atomic radius of 0.137 nm, and a c/a ratio of 1.615. Compute the volume of the unit cell for Re.
-
If the atomic radius of aluminum is 0.143 nm, calculate the volume of its unit cell in cubic meters.
-
Below is a unit cell for a hypothetical metal. (a) To which crystal system does this unit cell belong? (b) What would this crystal structure be called? (c) Calculate the density of the material,...
-
Kumara Corporation reported pretax book income of $1,530,000. Kumara also reports an increase in the taxable temporary differences of $327,500, an increase in the deductible temporary differences of...
-
Slumlord Floyd is getting a lot of complaints from tenants about how little care he takes of his rental property. Floyd ends up having several deductions disallowed under the Substandard Housing...
-
Nakashima Gallery had the following petty cash transactions in February of the current year. Nakashima uses the perpetual system to account for merchandise inventory. February 2 Wrote a $360 check to...

Study smarter with the SolutionInn App


