1. Rank the following compounds in order of increasing acidity. 2. Indicate which compounds would be more...
Question:
1. Rank the following compounds in order of increasing acidity.
2. Indicate which compounds would be more than 99% deprotonated by a solution of sodium ethoxide in ethanol.
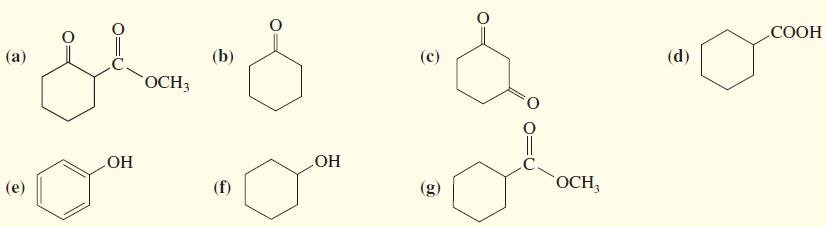
Transcribed Image Text:
СООН (а) (b) (с) (d) ОСН ОН ОН (е) (f) (g) OCH,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
In order of increasing acidity The most acidic protons are shown in b...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Rank the following compounds in order of increasing acidity. Dont look at a table Of PKa data to help with your answer. (a) Benzoic acid, p-methyl benzoic acid, p-chlorohenzoic acid (b)...
-
Rank the following compounds in order of decreasing acidity:
-
Rank the following compounds in order of increasing stability based on relative ring strain.
-
For the attractive-nuisance doctrine to apply, the possessor need not be aware that children are likely to trespass on his land or have reason to know that the condition poses an unreasonable risk of...
-
Harwell Company manufactures automobile tires. On July 15, 2011, the company sold 1,000 tires to the Nixon Car Company for $50 each. The terms of the sale were 2/10, n/30. Harwell uses the net method...
-
Rerun the new car simulation from Example 16.4, but now introduce uncertainty into the fixed development cost. Let it be triangularly distributed with parameters $600 million, $650 million, and $850...
-
In 2020, the Bureau of Labor Statistics posted the following statement to its website: Due to the unusual circumstances related to the pandemic . . . interviewers were given additional guidance...
-
The following is taken from Maxwell Co.s internal records of its factory with two operating departments. The cost driver for indirect labor and supplies is direct labor costs, and the cost driver for...
-
5 Which one of the following correctly represents Sodium oxide? +2 XX a) Na 20 XX 6) 2NaOx x; 1-2 'xx' 1-2 c) 2 Na 20% c) Na XX XX 6 An element with atomic number_ will form a basic oxide. a) 7 (2,5)...
-
What is the most correct IUPAC name for 2-(1-methylbutoxy)propane? 1-methyl-1-isopropoxybutane 2-isopropoxypentane 1-methyl-4-isopropoxybutane 2-oxypentylpropane 2-(oxymethylbutyl)propane
-
Propose a mechanism for the acid-catalyzed reaction of cyclohexanone with pyrrolidine.
-
Predict the products of the following aldol condensations. Show the products both before and after dehydration. (a) (b) (c) (d) (e) (f) CH3 TOH CH CH2-C-H CH TOH Ph-C-CH+ OH
-
Consider the matrix Use the determinant of A to state the conditions for which (a) A1 exists and (b) A1 = A. y A = 0 Z.
-
. Calculate the reaction forces at pins A and C, as well as the force exerted at pin B (a) without employing the concept of two-force members and (b) by utilizing the concept of two-force members (25...
-
It is now 23:20 hrs. You have conducted two 30 minute visual observations of Frieda - once at 22:30 hrs and again at 23:00 hrs. At 22:15hrs you say a quick hello and goodbye to the PCA from the...
-
The speed of sound in a classroom is 343 m/s. a) A tuning fork of frequency 512 Hz is struck. What length of open air tube is required to create a resonant sound at the 1 st harmonic? b) For the same...
-
a) What is the analogy of non-steady state diffusion of carburization and of decarburization in heat transport? b) Calculate the time necessary to obtain an 60 R, hardness effective case depth of 800...
-
Lisa is an ice skater. During a rotation, her initial angular velocity is 4 rev/s. She moves her arms in order to decrease her rotational inertia by 50%. Assuming the angular momentum of the system...
-
Name two areas where probability is used.
-
Two mutually exclusive investment alternatives are being considered. Alternative A requires an initial investment of $20,000 in a machine. Annual operating and maintenance costs are anticipated to be...
-
Starting with ketones and aldehydes of your choice, outline a directed aldol synthesis of each of the following using lithium enolates: (a) (b) (c) O OH CGH5 O OH
-
Assuming that dehydration occurs, write the structures of the two other products that might have resulted from the aldol cyclization just given. (One of these products will have a five-membered ring...
-
What starting compound would you use in an aldol cyclization to prepare each of the following? (a) (b) (c)
-
you retire with a large amount of savings in aretirement account. You decide to withdraw 3.5% to 4% of your savings the first year and increase the rate by inflation each year. What is the basis of...
-
test 1 for financial analysis and budgeting 1013 lambton college fpwt
-
Steed Company prepared a promissory note with a face value of $50,000, payable after 50 days, at a rate of 19%. Calculate the interest amount of the promissory note. Assume 365 days in a year. (Round...

Study smarter with the SolutionInn App


