A 13C NMR spectrum of commercially available 2, 4-pentanediol, shows five peaks at 23.3, 23.9, 46.5, 64.8,
Question:
A 13C NMR spectrum of commercially available 2, 4-pentanediol, shows five peaks at 23.3, 23.9, 46.5, 64.8, and 68.1 ?. Explain.
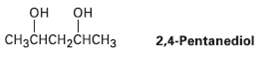
Transcribed Image Text:
он он CHзCнсH-CHCH3 2,4-Pentanediol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
Commercial 24pentanediol is a mixture of three stereoisomers RR SS and RS The meso isomer sh...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The mass spectrum and 13C NMR spectrum of a hydrocarbon are shown. Propose a structure for this hydrocarbon, and explain the spectraldata. 100 80 60 40 20 aly 120 10 40 60 80 100 140 m/z TMS 200 180...
-
A compound C6H14O has the 13C NMR spectrum shown in Figure 15.7. Its mass spectrum has a prominent peak at m/z 31. Suggest a reasonable structure for this compound.
-
A compound (C3H7ClO2) exhibited three peaks in its 13C NMR spectrum at 46.8 (CH2), 63.5 (CH2), and 72.0 ppm (CH). Excluding compounds that have Cl and OH on the same carbon, which are unstable, what...
-
Using the SEDAR database, find the most recent annual reports for two Canadian retailers (e.g., Loblaw, Rona, Danier Leather). Required: a. Based on the information provided in the companies audited...
-
In rapidly developing economies --- such as India and South Korea --- conglomerates are far more common than they are in the US and Western Europe. Use the BCG growth/share matrix to explain why this...
-
SUS Appliance toasters sell for $20 per unit, and the variable cost to produce them is $15. SUS estimates that the fixed costs are $80,000. a. Compute the break-even point in units. b. Fill in the...
-
Beginning with the score equations (11.4), verify the expression for the logit case in equation (11.5). a L() = x; (y; (x;)) i=1 '(x(B) (x)(1-(x)) = 0, (11.4)
-
Venture Camps, Inc., leases the land on which it builds camp sites. Venture is considering opening a new site on land that requires $2,500 of rental payment per month. The variable cost of providing...
-
On December 31, 2023, Green Bank enters into a debt restructuring agreement with Windsor Inc., which is now experiencing financial trouble. The bank agrees to restructure a $2.9-million, 10% note...
-
The file P02_18.xlsx contains daily values of the Standard & Poors 500 Index from 1970 to early 2012. It also contains percentage changes in the index from each day to the next. a. Create a histogram...
-
3-Methyl-2-butanol has five signals in its 13C NMR spectrum at 17.90, 18.15, 20.00, 35.05, and 72.75 ?. Why are the two methyl groups attached to C3 nonequivalent? Making a molecular model should be...
-
Carboxylic acids (RCO 2 H) react with alcohols (R?OH) in the presence of an acid catalyst. The reaction product of propanoic acid with methanol has the following spectroscopic properties. Propose a...
-
Graph the equation. 8x + 2y = -10
-
The volume of the tank is 12L and the time elapsed is non-negative. For which values of t does the equation V=4t accurately model the volume of the tank?
-
What are the methodological approaches used by symbolic interactionists in conducting research, such as participant observation, ethnography, and qualitative interviews, to explore the complexities...
-
Write the advantages and disadvantages your organization gained from internal and external sources of recruitment.
-
Side EF of square DEFG has a length of 2 cm and is also the radius of circle F. What is the area of the entire shaded region? Provide all evidence of your calculations. D 2 cm
-
What are the contemporary debates and revisions within modernization theory, considering the changing dynamics of globalization, digital technology, and environmental sustainability, and their impact...
-
Table 5.14 gives the yearly starting salaries for male and female bachelor's degree graduates who majored in the social sciences. a. Find and interpret a \(99 \%\) confidence interval for the...
-
The nitrogen atoms in N2 participate in multiple bonding, whereas those in hydrazine, N2H4, do not. (a) Draw Lewis structures for both molecules. (b) What is the hybridization of the nitrogen atoms...
-
Which molecule would you expect to be more soluble in water: CCl 4 or CH 2 Cl 2 ?
-
Show which of the nitrogen atoms in purine are basic, and which one is not basic. For the non-basic nitrogen, explain why its nonbonding electrons are not easily available to become protonated.
-
The proton NMR spectrum of 2-pyridone gives the chemical shifts shown. (a) Is 2-pyridone aromatic? (b) Use resonance forms to explain your answer to (a). Also explain why the protons at (7.31and...
-
Explain why each compound is aromatic, antiaromatic, or nonaromatic. (a) (b) (c) (d) (e) (f) (g) (h) N: isoxazole 1.3-thiazole pyran pyrylium ion y-pyrone 1.2-dihydropyridine NH2 N:
-
Lake Incorporated and the River, Incorporated reported net incomes of $205,000 and $164,000, respectively, for the most recent fiscal year. Both companies had 41,000 shares of common stock issued and...
-
What effect Eventually, BREXIT will make on EU? How it will benefit UK? What will be the possible outcome?
-
Merchant Company purchased property for a building site. The costs associated with the property were: Purchase price Real estate commissions Legal fees Expenses of clearing the land Expenses to...

Study smarter with the SolutionInn App


