A former theological student, Heavn Hardley, has turned to chemistry and, during his eighth year of graduate
Question:
A former theological student, Heavn Hardley, has turned to chemistry and, during his eighth year of graduate study, has carried out the following reaction:

Unfortunately, Hardley thinks he may have mislabeled his samples of A and 5, but has wisely decided to take an IR spectrum of each sample. The spectra are reproduced in Fig. P12.27 on p. 574. Which sample goes with which spectrum? How do you know?
Fig. P12.27
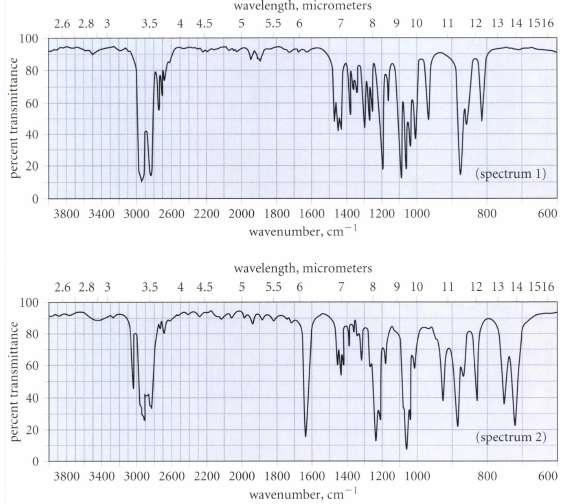
Transcribed Image Text:
Hz,catalyst wavelength, micrometers 2.6 2.8 3 3.5 4 4.55 5.5 6789 10 12 13 14 1516 100 E 80 60 C 40 20 (spectrum 1) 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 800 600 wavenumber, cm1 wavelength, micrometers 2.6 2.8 3 3.5 4.5 55.5 6 78 9 10 12 13 14 1516 100 80 E 60 ะ 40 o 20 0 (spectrum 2) 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 800 600 wavenumber, cm
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
Only spectrum 2 has the C C stretching absorption ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
A student carried out the following procedure to measure the pressure of carbon dioxide in a soft drink bottle. First, she weighed the bottle (853.5 g). Next, she carefully removed the cap to let the...
-
The following reaction is carried out at 500 K in a container equipped with a movable piston. After the reaction has reached equilibrium, the container has the composition depicted here. Suppose the...
-
A car owner who knows no chemistry has to put antifreeze in his car's radiator. The instructions recommend a mixture of 30% ethylene glycol and 70% water. Thinking he will improve his protection he...
-
The most common way of calculating finance charges is not the simplified one we used but rather the average daily balance. With this method, we calculate the account balance at the end of each day of...
-
Assume that for a 5-year period, large-company stocks had annual rates of return of 21.54 percent, -9.20 percent, -11.99 percent, -21.60 percent, and 29.39 percent. What is the variance of these...
-
Consider the relationship between a projects net present value (NPV), its internal rate of return (IRR), and a companys cost of capital. For each scenario that follows, indicate the relative value of...
-
Discuss the privacy and confidentiality issues of HIV-positive patients.
-
Big Bull restaurant employs twenty-three employees who receive tips. During the current year, Big Bull has $410,000 in gross revenues, and its employees do not report any tip income. In what ways may...
-
What strategies can be employed to enhance visibility and traceability across global supply chains, ensuring compliance with stringent regulatory frameworks and mitigating risks associated with...
-
Tony Anderson Productions is a sole trader-ship. On December 31, 2010 the following Trial Balance was extracted from the business's books: Trial Balance as at December 31, 2010 Details Return inwards...
-
Indicate how you would carry out each of the following chemical transformations. What are some of the changes in the infrared spectrum that could be used to indicate whether the reaction has...
-
Arrange the following bonds in order of increasing stretching frequencies, and explain your reasoning. C=C C=C C=0 C-C
-
Amanda Fall is starting up a new house painting business, Fall Colors. She has been advertising in the local newspaper for several months, and based on inquiries and informal surveys of the local...
-
As a renowned Project Management Consultant, discuss the characteristics/attributes that define a project and highlight the factors that should be considered when choosing a project selection model...
-
As a team leader, you make sure that each team member is encouraged to put in their best efforts and work towards a common goal. Discuss the following: In your experience, what makes the difference...
-
1. Where Do Leaders Learn to Lead? 2. Mention the six (6) Instrumental Values of Caring Leaders. 3. What are the 5 Traditional Leadership Duties vs the 5 New Demands of a Leader? 4.Mention five (5)...
-
1.What is project communication management and importance of communication in project management .please explain in detail 2. What are stakeholders and what is stakeholder engagement and why it is...
-
The border-adjustment feature of the House Republican tax plan is the key to cutting the corporate tax rateand stimulating economic growthwithout a major increase in the budget deficit. The tax would...
-
The management of Peoria Ltd, a US-based company, is reassessing the appropriateness of using its present inventory cost flow method, which is average cost. Management requests your help in...
-
The relationship described in question 7 does not always appear to hold. What factors, besides the number of firms in the market, might affect margins?
-
Draw the structures and give the names of all the dicarboxylic acids with the formula C 6 H 10 O 4 . Indicate which are chiral, which would readily form cyclic anhydrides on heating, and which would...
-
Draw the structure of the major species present in solution when 0.01 mole of the following acid in aqueous solution is treated with 0.01 mole of NaOH. Explain. O Cl O i OH HO-C-C-CH-CH-C-OH T CI
-
Give the product expected when butyric acid (or other compound indicated) reacts with each of the following reagents. (a) Ethanol (solvent), H 2 SO 4 catalyst (b) Aqueous NaOH solution (c) LiAlH 4...
-
Hall & Company issues $ 1 0 0 , 0 0 0 , 0 0 0 in 6 % bonds with detachable warrants of 1 0 warrants for each $ 1 , 0 0 0 bond. The bonds are issued at 9 9 . Record the issuance under the following...
-
Assume that hein acquired Dolan Development last year. Hein recorded the following intangible assets on the date of acquisition: Goodwill: $ 1 , 5 0 0 , 0 0 0 Dolan Development trademark: $ 6 0 0 , 0...
-
What is the total amount of interest that will be paid of the loan is carried to term with no extra payments made building cost 3 3 0 0 0 0 . loan started im Dec 2 0 2 3 worh a 7 . 5 % interest rate...

Study smarter with the SolutionInn App


