A fuel gas produced by gasifying coal is to be burned with 20% excess air. The gas
Question:
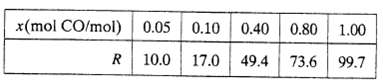
A fuel gas produced by gasifying coal is to be burned with 20% excess air. The gas contains 50.0 mole% nitrogen and the balance carbon monoxide and hydrogen. A sample of the gas is passed through an infrared spectrometer, which registers a signal R that depends on the mole fraction of carbon monoxide in the sample and a reading R 38.3 is recorded. Analyzer calibration data are as follows: A power law (x = aRb) should be suitable for fitting the calibration data. Derive the equation relating x and R (use a graphical method), and then calculate the molar flow rate of air required for a fuel feed rate of 175kmol/h, assuming that CO and H2 are oxidized but N2 is not.
Step by Step Answer:

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau





