A liquid mixture containing 70.0 mol of n -pentane and 30.0 mol of n -hexane initially at
Question:
A liquid mixture containing 70.0 mol of n-pentane and 30.0 mol of n-hexane initially at 46?C is partially vaporized at P = 1 atm in a single-stage distillation apparatus (Rayleigh still).
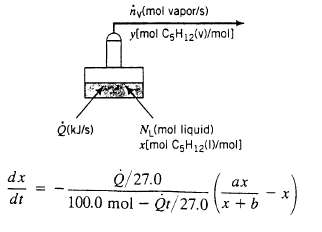
The heat added to the system, Q, vaporizes liquid at the rate nv (mol/s). The vapor product and remaining liquid at a given moment are always in equilibrium with each other. The relationship between the mole fraction of pentane in the liquid (x) and that in the vapor (y) is of the form y = ax/x + b so that the system involves four time-dependent variables?NL, nv, x, and y, where NL is the total moles of liquid in the still at any time. (We will suppose that the rate of heat transfer to the evaporator. Q is constant and known.) Four equations relating the unknowns will be required to determine these variables. The equations are two material balances, an energy balance, and the vapor?liquid equilibrium relationship just given.
(a) When x = 1, what must y equal? (Think of the definitions of these quantities.) Use your answer and the vapor?liquid equilibrium expression to derive an equation relating the parameters a and b.
(b) Use Raoult?s law (Equation 6.4-1) and the Antoine equation to calculate the mole fraction of pentane in the vapor phase in equilibrium with the 70% pentane?30% hexane feed mixture at the initial system temperature of 46?C and a pressure of 1 atm. Then use this result and that of part (a) to estimate a and b. (We will assume that these values remain the same over the range of compositions and temperatures to be undergone by the system.)
(c) Taking the residual liquid in the still as your system, write a differential balance on total moles to obtain an expression for dNL/dt. Then write a balance on pentane, recognizing that both L and x are functions of time. Prove that the pentane balance can be converted into the following equation: dx/dt = nv/NL (ax/x + b ? x) Supply initial conditions for your two differential equations.
(d) In part (c), you derived two equations in three unknown dependent variables??nv(t), NL (t), and x(t). To determine these variables, we need a third relationship. An energy balance provides it.
A rigorous energy balance would take into account the chancing composition of the liquid, the slightly different heats of vaporization of pentane and hexane and the enthalpy changes associated with temperature changes, and would make the problem relatively hard to solve. A reasonable approximation is to assume that (i) the liquid has a constant heat of vaporization of 27.0 kJ/mol. independent of composition and temperature; and (ii) all heat supplied to the still [Q(kJ/s)J goes to vaporize liquid (i.e., we neglect energy that goes into raising the temperature of the liquid or the vapor). Make these assumptions, consider Q to be constant and known, and derive a simple expression for nV that can be used to eliminate this variable in the differential equations of part (e). From there, derive the following expression:
(e) Use a differential equation-solving program to calculate x. y, NL, and nV from t = 0 until the time at which the liquid completely evaporates. Do the calculation for (i) Q = 1.5 kJ/s and
(ii) Q = 3.0 kJ/s, on a single graph, plot x and y versus r, showing curves for both values of Q.
(f) In a short paragraph describe what happens to the compositions of the vapor product and residual liquid over the course of a run. Include a statement of what the initial and final vapor compositions are and how the heating rate affects the system behavior.
Step by Step Answer:

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau





