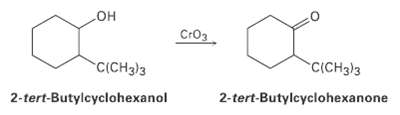
Alcohols undergo an oxidation reaction to yield carbonyl compounds on treatment with CrO 3 . For example,
Question:
Alcohols undergo an oxidation reaction to yield carbonyl compounds on treatment with CrO3. For example, 2-tert-butylcyclohexanol gives 2-tert-butylcyclo- hexanone. If axial ?OH groups are generally more reactive than their equatorial isomers, which do you think would react faster, the cis isomer of 2?tert?butylcvclohexanol or the trans isomer? Explain.
Transcribed Image Text:
HO Cro3 C(CH3)3 C(CH3)3 2-tert-Butylcyclohexanol 2-tert-Butylcyclohexanone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (18 reviews)
OH H OH CCH33 CCH33 cis trans OH OH H CCH33 H CCH33 All four conformat...View the full answer

Answered By

Lata Tiwari
I have been working as an Academic Consultant in the university.I have also experience of school teaching.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which factor do you think would have a larger effect on total fertility rate: the increased education of men or of women? Explain your answer.
-
Which medium (or media) do you think would be appropriate for each of the following kinds of messages a subordinate could receive from his or her boss: (a) A raise. (b) Not receiving a promotion. (c)...
-
Which do you think are the most important objectives?
-
With respect to strategies used by land conservation groups to preserve land, conservation easements seem to be expanding more rapidly than buying land for preservation. In what respect might...
-
Assume that at the beginning of the year, you purchase an investment for $7,000 that pays $100 annual income. Also assume the investment's value has decreased to $6,600 by the end of the year. a....
-
If the allowable tensile stress for wires AB and AC is Ï allow = 180 MPa, and wire AB has a diameter of 5 mm and AC has a diameter of 6 mm, determine the greatest force P that can be applied to...
-
Consider the Hald cement data in Table B.21. a. From the matrix of correlations between the regressors, would you suspect that multicollinearity is present? b. Calculate the variance inflation...
-
Unter Components manufactures low-cost navigation systems for installation in ride-sharing cars. It sells these systems to various car services that can customize them for their locale and business...
-
(13%) Problem 4: A student standing on a cliff throws a stone from a vertical height of d = 8.0 m above the level ground with velocity vo = 18 m/s at an angle 0 = 32 below the horizontal, as shown....
-
Snazzy Simis Swirls Simi has just completed her first year of university (most of her classes were related to business administration) and is now in the midst of making a major career decision. Since...
-
Ketones react with alcohols to yield products called acetals. Why does the allcis isomer of 4-tert-butyl-l, 3-cyclohexanediol react readily with acetone and an acid catalyst to form an acetal while...
-
Classify each of the following reactions as an addition, elimination, substitution, or rearrangement: (a) CH3Br + KOH CH3OH + KBr (b) CH3CH2Br H2C = CH2 + HBr (c) H2C = CH2 + H2 CH3CH3
-
In relation to internal controls, define: (i) A strength. (ii) A deficiency.
-
Generational issues (at least 3-4 pgs): What role are generational differences playing in the great resignation? Specifically, are there different rates of retention across generations within the...
-
Alignment of HR practices with company mission (3-4pgs): How well is Bank of America's compensation packages tied to performance management practices? Specifically, to what extent is Bank of...
-
HR Metrics (3-4 pgs with in-text citations (3-4) please): How are employers measuring the effectiveness of their retention strategies? In addition to actual retention statistics and comparison to...
-
You are the Manager for a local 24 hour pharmacy store. You hired Maria about two months ago as the Night Shift Lead. Overall her performance has been good. She has a good manner with customers, and...
-
Labor Unions answer the following questions: What are the advantages and disadvantages of labor unions? Are unions more popular or less popular now than they have been historically? Why?
-
Illustrate the following formulas with Euler diagrams, and give five concrete examples of each: P(A B) = P(A) + P(B) P(AB) and P(AB) = P(A) + P(B) P(A B).
-
Why is it necessary to study the diffusion of molecules in biological systems?
-
A group of spectroscopists believe that they have detected one of the following species: NeF, NeF + , or NeF - . Assume that the energy-level diagrams of Figure 11-25 apply, and describe bonding in...
-
Four benzenoid compounds, all with the formula C7H7Br, gave the following IR peaks in the 680-860-cm-1 region: A, 740 cm-1 (strong) B, 800 cm-1 (very strong) C, 680 cm-1 (strong) and 760 cm-1 (very...
-
Write structural formulas for each of the following: (a) 3-Nitrobenzoic acid (b) p-Bromotoluene (c) o-Dibromobenzene (d) m-Dinitrobenzene (e) 3,5-Dinitrophenol (f) p-Nitrobenzoic acid (g)...
-
Write structural formulas and give acceptable names for all representatives of the following: (a) Tribromobenzenes (b) Dichlorophenols (c) Nitroanilines (d) Methylbenzenesulfonic acids (e) Isomers of...
-
STS plc collects waste from houses, offices and factories. Most of the waste is burned to produce heat and electricity for the companys own use. This saves costs and reduces the impact on the...
-
Scenario You are the CFO of a Catholic School. You are asked to present recommendations to the board on two issues facing the school: First, the administration is considering purchasing a gymnasium...
-
A company projects an increase in net income of $30,000 each year for the next five years if it invests $300,000 in new equipment. The equipment has a five-year life and an estimated salvage value of...

Study smarter with the SolutionInn App


