Aromatic sulfonation can be reversed by hearting an arylsulfonic acid in steam: (a) Draw a curved-arrow mechanism
Question:
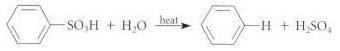
(a) Draw a curved-arrow mechanism for this transformation.
(b) Explain why compound C cannot be synthesized in one step from thiophene.
Transcribed Image Text:
-н + н.so,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
a By the principle of microscopic reversibility the mechanism for th...View the full answer

Answered By

Aun Ali
I am an Associate Member of Cost and Management Accountants of Pakistan with vast experience in the field of accounting and finance, including more than 17 years of teaching experience at university level. I have been teaching at both undergraduate and post graduate levels. My area of specialization is cost and management accounting but I have taught various subjects related to accounting and finance.
5.00+
13+ Reviews
32+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Treating oxytocin with certain reducing agents (e.g., sodium in liquid ammonia) brings about a single chemical change that can be reversed by air oxidation. What chemical changes are involved?
-
Monomethylation of histone Arg residues can be reversed by the action of a peptidylarginine deiminase, which requires water to remove the methyl group along with the imino group of Arg. Draw the...
-
Can a radical be aromatic?
-
Lewis and Associates has been in the termite inspection and treatment business for five years. The following is a list of accounts for Lewis on June 30, 2017. It reflects the recurring transactions...
-
a. Identify the main determinants for valuation of feature films, television programs, and general release feature productions by Columbia Pictures. b. Are the bases of valuation reasonable? Explain....
-
Wilbur Corporation is considering replacing a machine. The replacement will cut operating expenses by $24,000 per year for each of the five years the new machine is expected to last. Although the old...
-
Males Rating Females on OKCupid Exercise 2.188 introduced data showing the age of males that females find most attractive, based on the age of the females doing the rating. Here we examine the...
-
Remo Company and Angelo Inc. are separate companies that operate in the same industry. Following are variable costing income statements for the two companies showing their different cost structures:...
-
Here are the returns on two stocks: Digital Cheese Executive Fruit January +18 +8 February -4 +1 March +6 +5 April +8 +16 may -5 +2 June +4 +6 July -3 -4 August -9 -3 A-1) Calculate the variance...
-
Emerald City Umbrellas sells umbrellas and rain gear in Seattle, so its sales are fairly level across the year. However, it is branching out to other markets where it expects demand to be much more...
-
The dipole moments of pyrrole and pynolidine are similar in magnitude but have opposite directions. Explain, indicating the direction of the dipole moment in each compound. Should the dipole moment...
-
Provide curved-arrow mechanisms for each of the reactions given in Fig. P25.41. Give the structure for the intermediates A and B in parts (d) (a) (b) + DSo, (dilute) + HCI
-
The following model is a representation of acetaminophen, a pain reliever sold in drugstores as Tylenol. Identify the hydribization of each carbon atom in acetaminophen, and tell which atoms have...
-
New UK Prime Minister Liz Truss is reportedly weighing up scrapping the Soft Drinks Industry Levy (more commonly known as the sugar tax), in a move that could have knock-on effects for fizzy drinks...
-
What, in your opinion, are the three main problems, which could be studied through management sciences and entrepreneurship, and which the company faces, has faced or will face?
-
Explain financial options and how they are applied in corporate finance. ( with references )
-
Describe each step of the Database System Life Cycle and describe how they work together. Explain how the cycle can use in another context. For example, how can these steps be used when buying a car...
-
What are the two steps in the marketing process? Compare and contrast the product development process with the customer development process. What is the difference between the two and what are their...
-
Answer the following questions on ethics by conducting research using the library and/or internet and reflecting on your knowledge of ethics, your life experiences and beliefs. (a) What does the word...
-
Some people argue that the internal control requirements of the Sarbanes-Oxley Act (SOX) put U.S. companies at a competitive disadvantage to companies outside the United States. Discuss the...
-
How would you prepare the following ethers using a Williamson synthesis? (a) Methyl propyl ether (b) Anisole (methyl phenyl ether) (c) Benzyl isopropyl ether (d) Ethyl 2, 2-dimethylpropyl ether
-
Review the mechanism of oxymercuration shown in figure, and then write the mechanism of the alkoxy mercuration reaction of 1-methylcyclopentene with ethanol. Use curved arrows to show the electron...
-
How would you prepare the following ethers? Use whichever method you think is more appropriate, the Williamson synthesis or the alkoxy mercuration reaction. (a) Butyl cyclohexyl ether (b) Benzyl...
-
Show how binary exponentiation can be used to calculate a 128 efficiently. How many multiplications are required?
-
How can one tell many months the positive skew had in it?
-
Drug 4 mg I every 3 hours prn pain. Supplied as 10mg/10 ml. Calculate mL per dose?

Study smarter with the SolutionInn App


