At 20?C. 1,000 kg of a mixture of 50 wt% Na2S04 . 10H2O and 50 wt% Na2S04
Question:
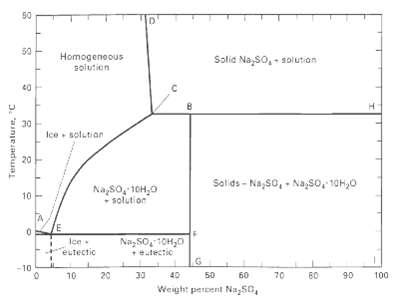
At 20?C. 1,000 kg of a mixture of 50 wt% Na2S04 . 10H2O and 50 wt% Na2S04 crystals exists. How many kilograms of water must be added to just completely dissolve the crystals if the temperature is kept at 20°C and equilibrium is maintained? UseFigure.
Transcribed Image Text:
50 50 Homogeneous solution Solia Na, so,- solution Ice solutian 20 Salids - Na,S0, - Na,So, 10H,0 Na,8o, 10H,0 solution 10 Na, SO, 10H,0 Ice eutectic +ealectio -10 60 10 20 30 40 50 60 70 90 100 Weight percent Na,50, Temperalure, "C 30
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
From Fig the solubility of Na 2 SO 4 in water at 20 o C is 15 wt ...View the full answer

Answered By

Jeff Omollo
As an educator I have had the opportunity to work with students of all ages and backgrounds. Throughout my career, I have developed a teaching style that encourages student engagement and promotes active learning. My education and tutoring skills has enabled me to empower students to become lifelong learners.
5.00+
5+ Reviews
42+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
How many kilograms of nickel must be added to 1.75 kg of copper to yield a liquids temperature of 1300(C?
-
How many kilograms of nickel must be added to 5.43 kg of copper to yield a solidus temperature of 1200(C?
-
How many grams of MgO must be added to 1 kg of NiO to produce a ceramic that has a solidus temperature of 2200 C?
-
Why is it likely to have preexisting normal faults in an orogenic belt?
-
Tarika Ltd. is a profitable small business. It has not, however, given much consideration to internal control. For example, in an attempt to keep clerical and office expenses to a minimum, the...
-
Timpanogos Inc. is an accrual-method calendar-year corporation. For 2020, it reported financial statement income after taxes of $1,342,000. Timpanogos provided the following information relating to...
-
A certain 10-year bond is currently selling for \(\$ 920\). A friend of yours owns a forward contract on this bond that has a delivery date in 1 year and a delivery price of \(\$ 940\). The bond pays...
-
Tidy House produces a variety of household products. The firm operates 24 hours per day with three daily work shifts. The first-shift workers receive regular pay. The second shift receives an 8...
-
Select a metallic ship or boat component that is manufactured starting from a billet using at least two combinations of forging, rolling, extrusion or wire drawing (ex. Hot extrusion then forging). ...
-
Each barbecue is sold for $5.00, and each balloon is sold for $2.00. Throughout the two weekend days, the four friends were able to sell 300 barbecues and 170 balloons. The four friends did not have...
-
Repeat Example 4.11, except let the original solution be 20 wt% Na2S04.
-
Repeat Example 4.12, except determine the grams of activated carbon to achieve: (a) 75% adsorption of phenol. (b) 90% adsorption of phenol. (c) 98% adsorption of phenol.
-
Economists call the increase in insurance costs that resulted from the September 11 th attacks an _____ shock which leads to the ________. a. Aggregate demand; aggregate demand curve shifting left b....
-
For some records, the record temperature occurred more than once. In these cases, only the most recent year is listed. Explain how this might cause the mean to be greater than the midpoint of 1945.5...
-
True or False? The base case is what allows recursion to terminate.
-
The distance advanced in a day by a tunnel boring machine, in meters, is uniformly distributed on the inter\(\operatorname{val}(30,50)\). a. Find the mean distance. b. Find the standard deviation of...
-
Write a Copy member function of the Stack ADT, assuming that self is copied into the stack named in the parameter list.
-
Of the three variations of linked lists (circular, with header and trailer nodes, and doubly linked), which would be most appropriate for each of the following applications? 1. You want to search a...
-
Bingo Oil Corporation recently acquired a truck costing $60,000 with an estimated life of five years (ignore salvage value). The foreman drives the truck to oversee operations on seven leases, all in...
-
In the synthesis of the keto acid just given, the dicarboxylic acid decarboxylates in a specific way; it gives Explain. HO rather than HO
-
Explain what is meant by push and pull factors in relation to turnover. What can an organization do, if anything, to control these factors?
-
What is diafiltration? How does it differ from continuous feed-and-bleed ultrafiltration? Under what conditions is diafiltration used in conjunction with continuous feed-and-bleed ultrafiltration?
-
In Example 8.19, the % extraction of lysozyme is high (95.2%), but the % purity of the lysozyme in the total extracted proteins is low (15.1%). The Kremser plot of Figure 5.9 suggests that by...
-
What is continuous feed-and-bleed ultrafiltration? What are its limitations?
-
There are 42 locations throughout the northeast region with deposits totaling $3.2bil. If you were Kiamarie's vice president, how would you expect her to choose between Gagne's Nine Events of...
-
Why is succession planning essential in an organization? 2. What is the need for succession planning? 3. What do you think are the challenges to succession planning? 4. How can employees contribute...
-
Two activities/obligations/responsibilities that might be added to an updated version of the job description for an HR professional: How would you go about monitoring changes in laws and regulatory...

Study smarter with the SolutionInn App


