Below is a series of cost of goods sold sections for companies B, F, L, and R.
Question:
Below is a series of cost of goods sold sections for companies B, F, L, and R.
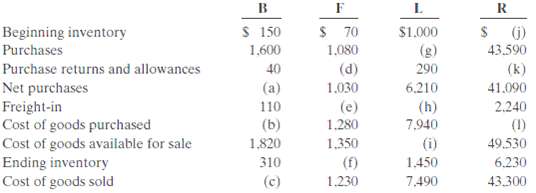
InstructionsFill in the lettered blanks to complete the cost of goods soldsections.
Transcribed Image Text:
Beginning inventory $ 150 $ 70 $1.000 (g) (j) 43.590 1.600 40 (a) 110 (b) 1,820 310 (c) 1,080 Purchase returns and allowances Net purchases Freight-in 290 6.210 (h) 7,940 (i) 1.450 7,490 (d) (k) 41.090 2.240 (1) 49.530 6.230 43.300 1.030 (e) 1,280 Cost of goods purchased Cost of goods available for sale Ending inventory Cost of goods sold 1,350 (f) 1.230
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
a 1560 1600 40 b 1670 1560 110 c 1510 1820 310 ...View the full answer

Answered By

Mishark muli
Having any assignments and any other research related work? worry less for I am ready to help you with any task. I am quality oriented and dedicated always to produce good and presentable work for the client once he/she entrusts me with their work. i guarantee also non plagiarized work and well researched work to give you straight As in all your units.Feel free to consult me for any help and you will never regret
4.70+
11+ Reviews
37+ Question Solved
Related Book For 

Accounting Principles
ISBN: 978-0470533475
9th Edition
Authors: Jerry J. Weygandt, Paul D. Kimmel, Donald E. Kieso
Question Posted:
Students also viewed these Accounting questions
-
Below is the cost of goods sold sections for the two most recent years for two companies using a periodic inventory system: Instructions Fill in the missing amounts to complete the cost of goods sold...
-
Below are the cost of goods sold sections for the most recent two years for two companies using a periodic inventory system: Instructions Fill in the numbered blanks to complete the cost of goods...
-
The following is a series of annual sales (in $ millions) over an 11- year period ( 2003 to 2013): a. Construct a time- series plot. b. Does there appear to be any change in annual sales over time?...
-
In a given week, 12 babies are born in hospital. Assume that this sample came from an underlying normal population. The length of each baby is routinely measured and is listed below (in cm): 49, 50,...
-
A helicopter moves horizontally in the x direction at a speed of 120 mi/h. Knowing that the main blades rotate clockwise with an angular velocity of 180 rpm, determine the instantaneous axis of...
-
Discuss some of the techniques used to cut fiber-reinforced composites.
-
A loop of wire carries a current \(l\) as shown in Figure P28.13. Determine the direction of the magnetic field at the points labeled. Data from Figure P28.13 L x 2 3
-
Megley Cheese Company is a small manufacturer of several different cheese products. One of the products is a cheese spread that is sold to retail outlets. Jason Megley must decide how many cases of...
-
Question four (20mks) Explain how the following are fundamental in appraising income property A. potential gross income (16mks) B.Net operating income (4mks) Question five (20mks) Explain the theory...
-
Consider a particle is confined in a three-level system. The probability of finding the particle in first level is 0.38, for second level 0.36, and for third level it is 0.26. What is the entropy of...
-
On January 1, 2010, Rachael Ray Corporation had merchandise inventory of $50,000. At December 31, 2010, Rachael Ray had the following account balances. Freight-in........... $ 4,000...
-
This information relates to Martinez Co. 1. On April 5 purchased merchandise from D. Norlan Company for $20,000, terms 2/10, net/30, FOB shipping point. 2. On April 6 paid freight costs of $900 on...
-
What is sustainability? How might the firm undertake sustainability in its value-chain activities?
-
Beagle Beauties engages in the development, manufacture, and sale of a line of cosmetics designed to make your dog look glamorous. Below you will find selected information necessary to compute some...
-
Explain some of the reasons why developing countries have not had a greater positive impact on the development of their higher education programs. In this context, why should the development of a...
-
Solve the equation: 5t = 5t +2
-
Which of the following is caused by a point mutation that causes a change in the amino acid during translation? Hereditary spherocytosis thalassemia Sickle cell anemia Cystic fibrosis With an...
-
Apollo Industries' main product is gondola cars for amusement parks and ski lifts. Your marketing department constantly monitors trends in the external environment that may affect the company. What...
-
The following data are available for Retique! Net cash provided by financing activities is: a. \(\$ 90,000\). b. \(\$ 130,000\). c. \(\$ 160,000\). d. \(\$ 170,000\). Increase in accounts payable...
-
Using the information in P11-2B, compute the overhead controllable variance and the overhead volume variance. Data From Problem 11-2B: Huang Company uses a standard cost accounting system to account...
-
Draw dot diagrams for the following simple ions: (a) I (b) O 2 (c) Cl (d) H +
-
Prior Company buys merchandise on account from Wood Company. The selling price of the goods is $900 and the cost of the goods sold is $630. Both companies use perpetual inventory systems. Journalize...
-
Prepare the journal entries to record the following transactions on Ramirez Companys books using a perpetual inventory system. (a) On March 2 Ramirez Company sold $800,000 of merchandise to Ikerd...
-
From the information in BE5-3, prepare the journal entries to record these transactions on Ikerd Companys books under a perpetual inventory system.
-
Regarding the following structure answer the following questions: N 1) What is the sar? wwww olaparib (lynparza) 2)What is the quantitive estimation (principle of assay)?
-
7. Determine the Q-values of the following reaction: N+13N P+13C (Given: Rest-mass of 13N = 13.005739 u; rest-mass of 13C = 13.003355 u.) (3 marks)
-
Consider the following description for the element iron, Fe Ph Iron is a silver-white malleable ductile magnetic heavy metallic element that readily rusts in moist air to give a hydrated iron oxide....

Study smarter with the SolutionInn App


