Compounds A and B are isomers with the formula C3H6O. A has a peak at 1730 cm
Question:
Compounds A and B are isomers with the formula C3H6O. A has a peak at 1730 cm?1 in its IR spectrum and B has a peak at 1715cm?1, the mass spectra of A and B are as follows, show the structures of A and B.
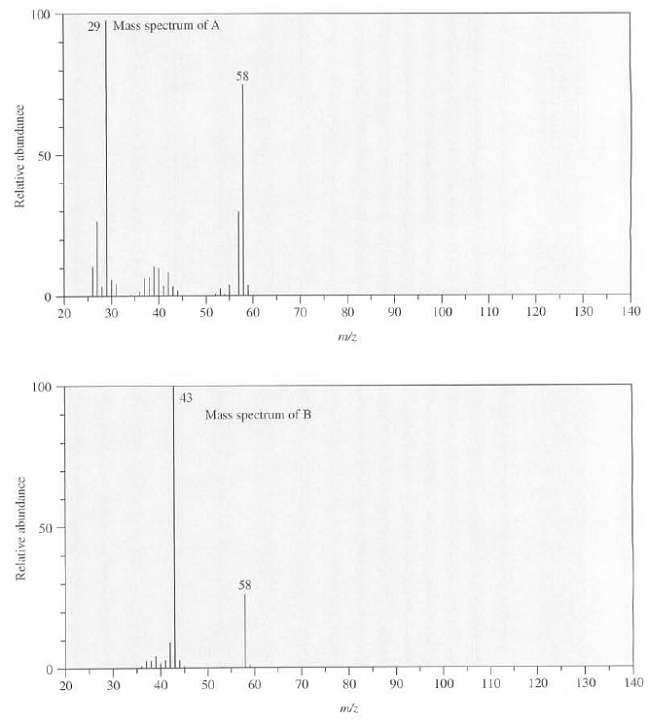
Transcribed Image Text:
100 29 | Mass spectrum of A 58 50 140 70 80 90 100 110 120 130 20 30 40 50 60 100 43 Mass spectrum of B 50 58 140 120 130 70 80 90 100 110 30 40 50 60 20 Relative abundance Relative abundance
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
The DU for both isomers is 1 Based on their PR spectra this must result from a carbonyl group ...View the full answer

Answered By

Gabriela Rosalía Castro
I have worked with very different types of students, from little kids to bussines men and women. I have thaught at universities, schools, but mostly in private sessions for specialized purpuses. Sometimes I tutored kids that needed help with their classes at school, some others were high school or college students that needed to prepare for an exam to study abroud. Currently I'm teaching bussiness English for people in bussiness positions that want to improve their skills, and preparing and ex-student to pass a standarized test to study in the UK.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Compounds A and B are isomers having the molecular formula C4H8O3. Identify A and B on the basis of their 1H NMR spectra. Compound A: 1.3 ppm (3H, triplet); 3.6 ppm (2H, quartet); 4.1 ppm (2H,...
-
Compounds A and B are isomers of molecular formula C10H14. Identify each one on the basis of the 13C NMR spectra presented in Figure 13.41.
-
Compounds A, B, and C are isomers with the formula C5H11Br. Their broadband proton-decoupled 13C NMR spectra are given in Fig. 9.32. Information from the DEPT 13C NMR spectra is given near each peak....
-
At Blossom Company, events and transactions during 2020 included the following. The tax rate for all items is 20%. (1) Depreciation for 2018 was found to be understated by $148000. (2) A strike by...
-
How does administrative responsibility contribute to the attainment of public interest?
-
What additional information do you need about the downsizing effort at Brooks to fully understand the legal implications of its restructuring?
-
Find a recent sustainability report. Good reports can be found at the Australian Reporting Awards website; however, it might be useful to compare these to other firms reports. Required Prepare a...
-
Firm S is the only producer of a particular type of foam fire retardant and insulation used in the construction of commercial buildings. The inverse demand equation for the product is P = 1,500 -...
-
1. In the system shown in figure, all surfaces are smooth. Rod is moved by external agent with acceleration 0.9 ms vertically downwards. Force exerted on the rod by the wedge will be Newtons.
-
A Global private bank is aggressively looking to leverage technology to improve customer experience and reduce operational costs. Over the last few years, it has tied up with at least five startups...
-
The mass spectra of 3-ethyl-2-pentanone and 4-methyl-2-pentanone are as follows. Explain which spectrum goes with which compound, what is the structure of the ion responsible for the peak at m/z 43...
-
Compounds C and D are isomers with the formula C9H12, in addition to other absorption peaks, both compounds show a peak near 7.25 ? (area 5) in their 1H-NMR spectra. Their mass spectra are as follow,...
-
Billie Whitehorse, the plant manager of Travel Free's Ohio plant, is responsible for all of that plant's costs other than her own salary. The plant has two operating departments and one service...
-
During 2 0 2 5 , Wildhorse Corp. entered into the following transactions. 1 . Borrowed $ 6 1 , 0 0 0 by issuing bonds. 2 . Paid $ 9 , 3 5 0 cash dividend to stockholders. 3 . Received $ 1 8 , 8 0 0...
-
"Municipalities in South Africa are faced with a serious challenge of service delivery. It is argued that in some instances this emanates from poor talent management strategies that fail to retain...
-
Brothers Flooring Two brothers, Ali and Abbas Hamdouni have started a flooring business installing tiles, laminate and hardwood floors in residential units. From their start they have been very keen...
-
A Zero-Coupon bond has a par value equal to $1,200.The bond will mature in 15 years and has a yield to maturity of 6%.If we assume annual coupon payments, what was the bond's original issue price? PV...
-
What to use as "S". The directions say "The negative NPV of the initial investment as the price of the option". Would this mean the present value of the 200 and 250 million dollar investments, or the...
-
In 2018 New Zealand rewrote the charter of its central bank to include high employment as well as low inflation as its goals. Why would New Zealand want to do this?
-
What kind of rays are X-rays?
-
Three economists at the Federal Reserve Bank of St. Louis Andrew Levin, Fabio Nattaluci, and Jeremy Piger have attempted to measure the effects of a short-lived increase in actual inflation on...
-
Write structures for all compounds with molecular formula C4H6O that would not be expected to exhibit infrared absorption in the 3200-3550-cm-1 and 1620-1780-cm-1 regions.
-
Add curved arrows to the following reactions to indicate the flow of electrons for all of the bond-forming and bond-breaking steps. (a) (b) H.
-
Most carboxylic acids dissolve in aqueous solutions of sodium bicarbonate (NaHCO3) because, as carboxylate salts, they are more polar. Write curved arrows showing the reaction between a generic...
-
Describe and analyze issues in of subordination and privilege, oppression and resistance in their own lives and the lives of other cultural groups related to sports ?
-
Analyze each transaction. Under each category in the accounting equation, indicate whether the transaction: A. increases, B. decreases, or C. has no effect. The item (a) is provided as an example....
-
A motorist travels 70 mi while driving in a bad rainstorm. In sunny weather, the motorist drives 30 mph faster and covers 130 mi in the same amount of time. Find the speed of the motorist in the...

Study smarter with the SolutionInn App


