Into how many peaks would you expect the 1H NMR signals of the indicated protons to be
Question:
Into how many peaks would you expect the 1H NMR signals of the indicated protons to be split? (Yellow-green =Cl.)
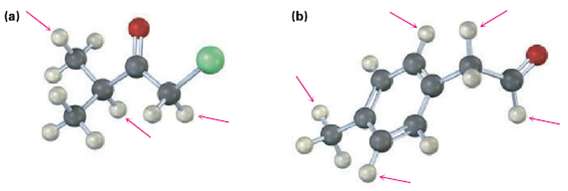
Transcribed Image Text:
(b) (a)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (19 reviews)
a HC H3C H 00 CHCl 1 dou...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How many peaks would you expect to see in the 1H-decoupled 13C NMR spectrum of a. 2-methyl-2-propanol? b. 4-methylcyclohexanone? c. 2-methyl-1-propanol? d. cis-1,3-dimethylcyclopentane?
-
How many peaks would you expect in the 1H NMR spectrum of 1, 4-dimethyl-benzene para-xy1ene or p-xylene)? What ratio of peak areas would you expect on integration of the spectrum? Refer to Table 13.3...
-
How many 1H NMR signals would you expect from each of the following compounds? (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) (m) (n) (o) (p) OH CI
-
In a recombinant protein expression process the host microorganism, Pichia pastoris needs to be separated from the culture supernatant after production. A pilot scale bioreactor with a working volume...
-
"The only way to succeed in a market with homogeneous products is to produce more efficiently than most firms." Comment. Does this imply that efficiency is less important in oligopoly and monopoly...
-
Consider two firms, one with an FF rating and one with an FFF rating. What is the probability that after 4 years each will have retained its rating? What is the probability that each will have moved...
-
A neutron star is an object with a mass of about \(1.4 M_{\odot}\), where the solar mass \(M_{\odot}=2 \times 10^{30}\) \(\mathrm{kg}\). The neutron star is aggregated to have density equal to that...
-
On January 1, 2014, Alter Company had Accounts Receivable $154,000; Notes Receivable of $12,000; and Allowance for Doubtful Accounts of $13,200. The note receivable is from Hartwig Company. It is a...
-
(a) X Ltd. is studying the possible acquisition of Y Ltd. by way of merger. The following data are available in respect of both the companies. Particulars Market Capitalization (Rs.) Gross Profit...
-
You are a senior auditor with Rodriguez & Jones, a small auditing firm located in Canterbury, an eastern suburb of Melbourne, Victoria. Your team has been assigned to the audit of a new client,...
-
How could you use 1H NMR to determine the regiochemistry of electrophilic addition to alkenes? For example, does addition of HCI to 1-methylcyclohexene yield 1-chloro-1-methylcyclohexane or...
-
How many absorptions would you expect the following compound to have in its 1H and 13C NMRspectra?
-
The auditors sometimes decide to allocate the amount of planning materiality to various financial statement accounts. a. Explain why auditors typically decide to allocate planning materiality to...
-
Improve the organization, coherence, and correctness of the following paragraph. We feel that the extreme strategy has not been developed fully in the fast-food market. Pizza Hut is considering...
-
Which cloud computing model offers the most control of your environment? A. PaaS B. SaaS C. IaaS D. DBaaS
-
A prospective client in Italy wants price quotes for a number of your productspronto! Using Figure 2.5, suggest the best communication channels for the following messages. Assume that all channels...
-
Where are the unrealized foreign currency gains and losses put in a hedging relationship?
-
Which sentence should be the topic sentence? Your Task. Study the following list of sentences from an interoffice memo to hospital staff. 1. The old incident report form caused numerous problems and...
-
In addition to full cost accounting and differential accounting, a third type of accounting information is used in the management control process. (Anthony and Reece). What is this third type of...
-
Cassandra Casey operates the Futuristic Antique Store. She maintains subsidiary ledgers for accounts payable and accounts receivable. She presents you with the following information for October 2019:...
-
A solution contains 4.08 g of chloroform (CHCl 3 ) and 9.29 g of acetone (CH 3 COCH 3 ). The vapor pressures at 35 C of pure chloroform and pure acetone are 295 torr and 332 torr, respectively....
-
The NMR spectrum of cinnamaldehyde follows. (a) Determine the chemical shifts of Ha, Hb and Hc. The absorption of one of these protons is difficult to see; look carefully at the integrals. (b)...
-
Consider the proton NMR spectrum of the following ketone. (a) Predict the approximate chemical shift of each type of proton. (b) Predict the number of NMR peaks for each type of proton. (c) Raw a...
-
Use the imaginary replacement technique to show that protons Hc and Hd in cyclobutanol are diastereotopic.
-
In Unix and Linux systems, the configuration of the operating system, servers, applications and preferences is commonly stored in plain text files. You want to view the hardware attached to the...
-
An actual corporation reported this information on their 2019 Annual Report ($ millions). Inventory cost method of LIFO is used. Date Inventory Balance Cost of Goods Sold (COGS) December 31, 2018...
-
The markets in general are paying a 2% real rate of return. Inflation is expected to be 3%. ABC stock commands a 6% risk premium. What is the expected rate of return on ABC stock? The required rate...

Study smarter with the SolutionInn App


