On reaction with Cl 2 in the presence of light, an unknown compound with the formula C
Question:
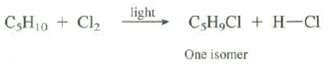
On reaction with Cl2 in the presence of light, an unknown compound with the formula C5H10 gives only one isomer of C5H9Cl (see problem 2.39). What is the DU of the unknown compound? Show the structure of the unknown compound and the product of its reaction with Cl2.
Transcribed Image Text:
CsH₁0 + Cl₂ light C,H,Cl + H-CI One isomer
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
The DU of C5H0 25 2 122 1 so the unknown has one rin...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
An unknown compound of molecular formula C5H9NO gives the IR and NMR spectra shown here. The broad NMR peak at δ7.55 disappears when the sample is shaken with D2O. Propose a structure,...
-
On heating 1,2,4-butanetriol in the presence of an acid catalyst, a cyclic ether of molecular formula C4H8O2 was obtained in 81-88% yield. Suggest a reasonable structure for this product.
-
In the presence of an acid catalyst, acetaldehyde forms a trimer known as paraldehyde. Because it induces sleep when it is administered to animals in large doses, paraldehyde is used as a sedative or...
-
Marcus is the HR manager for United Airlines, an Illinois-based company. One of his employees has recently become disabled and is unable to fulfill the essential functions of his current position,...
-
What are the advantages and disadvantages of basing individual incentives on company-wide performance?
-
If she uses the new paint, by what amount could Ms. French's material costs increase without reducing total (multifactor) productivity?
-
Comment on the statement: Classes provide the containers and objects provide the contents.
-
Determine the force in members EI and JI of the truss which serves to support the deck of a bridge. State if these members are in tension or compression. Units Used: kip = 103 lb Given: F1 = 4000 lb...
-
HomeLife Life Insurance Company has two service departments (actuarial and premium rating) and two production departments (advertising and sales). The distribution of each service department's...
-
Which of the graphs in Fig. Q25.12 best illustrates the current I in a real resistor as a function of the potential difference V across it? Explain. Figure Q25.12 (a) (b) (c) (d)
-
One of the isomers of C 5 H 12 reacts with Cl 2 in the presence of light to produce three isomers of C 5 H 11 Cl: This reaction replaces am one of the hydrogen?s of C 5 H 12 with a Cl. What arc the...
-
Explain how the dipole moment for CH3Cl ( = 1.9 D) can be larger than the dipole moment for CH3F ( = 1.8 D).
-
What is meant by the term the limitations of the research, in the context of the final chapter of the thesis or report of the research?
-
About 30% of the forests in Japan are managed by the government as national forests. Do you think they should be privatized? Why or why not?
-
(a) An amount of substance equal to 2 kmols of an ideal gas at temperature \(T\) and pressure \(p\) is contained in a compartment. In an adjacent compartment is an amount of substance equal to \(1...
-
The heat of reaction of methane \(\left(\mathrm{CH}_{4} ight)\) is determined in a constant pressure calorimeter by burning the gas as a very weak mixture. The gas flow rate is \(70 \mathrm{~L} /...
-
Calculate the lower heat of reaction at constant volume for benzene \(\mathrm{C}_{6} \mathrm{H}_{6}\) at \(25^{\circ} \mathrm{C}\). The heats of formation at \(25^{\circ} \mathrm{C}\) are: benzene,...
-
The products of combustion of a jet engine have a molecular weight, \(m_{w}\), of 30 and a molar specific heat at constant pressure given by \(c_{p, m}=3.3 \times 10^{4}+15 T \mathrm{~J} /...
-
From Barack and Michele Obama to Bill and Melinda Gates, to Brad Pitt and Angelina Jolie, romance befalls many at work, whether they are working at a law firm or a software company, or making a movie...
-
Draw two scatterplots, one for which r = 1 and a second for which r = 21.
-
A force of 40 N is required to hold a spring that has been stretched from its natural length of 10 cm to a length of 15 cm. How much work is done in stretching the spring from 15 cm to 18 cm?
-
Rank the following substances in order of increasing acidity: (a) (CH3)2CHOH, HC CH, (CF3)2CHOH, CH4OH (b) Phenol, p-methyl phenol, p-(trifluoromethyl) phenol (c) Benzyl alcohol, phenol, p-hydroxy...
-
P-Nitro benzyl alcohol is more acidic than benzyl alcohol but p-methoxy benzyl alcohol is less acidic. Explain.
-
Predict the products of the following reactions: CH (a) 1. CHCH2- 2. NaOH, H2O2 "CH (b) 1. HglOAc)2. 0 2. NABH4 (c) CCH2CH2CH2 CH2CH2CH2CH3 C=C 1. Os04 2. NaHSO3, H20 -
-
How does the central bank imply Bank Rate fluctuations as the monetary policy mechanism? Explain.
-
1. Calculate the NPV and Profitability Index for each of the following projects, assuming the cost of capital is 7%. YEAR iPad Pro iPhone 9 I 0 -36,000 -11,500 1 18,000 0 2 12,600 7,500 3 4,500 4,000...
-
State the dividend irrelevance proposition. What are the assumptions behind this proposition? Explain why this proposition does not hold in the real world. (20 marks)

Study smarter with the SolutionInn App


