Predict the aldol reaction product of the followingcompounds: (c) (b) (a) CCH-CH2CH CH3
Question:
Predict the aldol reaction product of the followingcompounds:
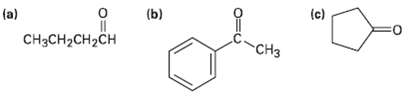
Transcribed Image Text:
(c) (b) (a) CнзCH-CH2CH CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (20 reviews)
1 Form the enolate of one molecule of the carbonyl compound CH3CHCHICH OH CH3CHCHCH 2 ...View the full answer

Answered By

Dinesh F
I have over 3 years of professional experience as an assignment tutor, and 1 year as a tutor trainee.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the products from each of the following aldol reactions. (a) (b) (c) (d) (e) H NaOH H20 H H NaOH H2O o'H H HOEN NaOH H2O H NaOH H2O
-
Predict the products of the following aldol condensations. Show the products both before and after dehydration. (a) (b) (c) (d) (e) (f) CH3 TOH CH CH2-C-H CH TOH Ph-C-CH+ OH
-
The following reaction does not produce the product shown. (a) Predict the major product from the conditions shown above, and write a detailed mechanism for its formation. (b) What reaction...
-
Neer Department Store uses the retail inventory method to estimate its monthly ending inventories. The following information is available for two of its departments at August 31, 2011. Sporting Goods...
-
Your client is the planning office of a major university. Part of the job of the planning office is to forecast the annual donations of alumni through the university's long-established giving...
-
The response of a system to a unit step change in the input (occurring at time 0) is shown in Fig. E7.11. (a) Derive a second-order plus time delay model approximation for the system. Provide values...
-
Describe how human genetics and stem cell research can have an impact on end-of-life issues.
-
On December 31, 2006 Marsh Company held 1,000 shares of X Company common stock in its portfolio of long-term investments in available-for-sale securities. The stock had cost $15 per share and has a...
-
What measures can be implemented to address the growing cybersecurity threats targeting supply chain infrastructure, safeguarding critical assets and sensitive information against data breaches and...
-
Use the Backtracking algorithm for the m-Coloring problem (Algorithm 5.5) to find all possible colorings of the graph in the attached file using the three colors red, green, and white. Show the...
-
Carboxylic acids (RCO 2 H; p K a 5) are approximately 10 11 times more acidic than alcohols (ROH; pK a 16). In other words, a carboxylate ion (RCO 2 ) is more stable than an alkoxide ion (RO )....
-
Using curved arrows to indicate the election flow in each step, show how the base-catalyzed reverse aldol reaction of 4-hydroxy-4-methly-2-pentanone takes place to yield 2 equivalents of acetone.
-
Give the differential equation that models exponential growth and decay.
-
Aster invested $790 for 5 years. At the investment's maturity, its value was $1090.20. What was the annual simple interest rate?
-
Kody was expelled from high school for getting explosively angry at a teacher who according to Kody, had humiliated him in front of his classKody was told that he would not be readmitted to school...
-
Briefly describe a finance and mortgage broker's obligations under the best interests duty. in 40 words
-
There are 4 types of analysis of financial statements-Explaining in your own words what isHorizontal Analysis.
-
You have a 6-year-old bilingual second language learner on your caseload who has an IEP. He was diagnosed as speech/language impaired by a previous therapist, and has goals to work on: increasing...
-
Locate at least one article that comments on the tax provisions for capital gains that were included as part of the 2012 Taxpayer Relief Act. Summarize the comments and provide a citation for your...
-
AB CORPORATION ISSUED THE FOLLOWING 850 COMMON STOCKS PAR VALUE P100 750 PARTICIPATING PREFERRED STOCKS PAR VALUE P100 AT 3% AB CORPORATION DECLARED P100,000.00 DIVIDEND IN 2022.
-
Draw the Lewis structure for HCSNH 2 . (The carbon and nitrogen atoms are bonded together, and the sulfur atom is bonded to the carbon atom.) Label each bond in the molecule as polar or nonpolar.
-
Calculate the specific rotations of the following samples taken at 25 C using the sodium D line. (a) 1.00 g of sample is dissolved in 20.0 mL of ethanol. Then 5.00 mL of this solution is placed in a...
-
Tartaric acid has a specific rotation of +12.0o. Calculate the specific rotation of a mixture of 68% (+)-tartaric acid and 32% (-) tartaric acid.
-
The specific rotation of (S)-2-iodobutane is +15.90o. (a) Draw the structure of (S)-2-iodobutane. (b) Predict the specific rotation of (R)-2-iodobutane. (c) Determine the percentage composition of a...
-
There are peer effects, but they differ for the two individuals, u_{1} = x_{1} - x_{1} ^ 2 + beta_{1}*x_{1}*x_{2} with B_{1} = 1 U_{2} = O - x_{2} ^ 2 + beta_{2}*x_{1}*x_{2} with B_{2} = 0 1) Find...
-
How do emerging technologies, such as AI and data analytics, impact the evolution of performance management practices ?
-
The controller for Rainbow Children's Hospital, located in Munich, Germany, estimates that the hospital uses 38 kilowatt-hours of electricity per patient-day, and that the electric rate will be 0.26...

Study smarter with the SolutionInn App


