Propose structures for compounds with the following 1H NMR spectra: (a) C 5 H 9 ClO 2
Question:
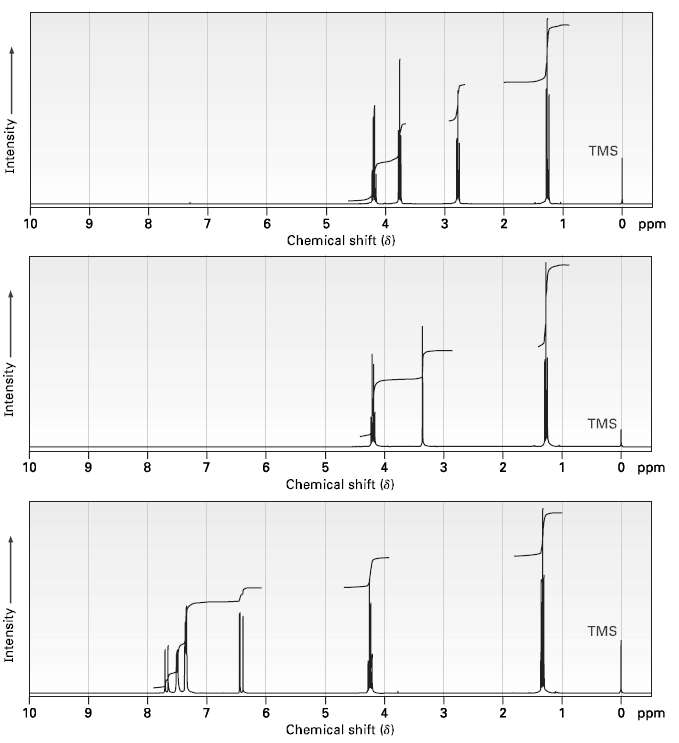
Propose structures for compounds with the following 1H NMR spectra:
(a) C5H9ClO2? ?IR: 1735 cm?1?
(b) C7H12O4? ? ?IR: 1735 cm?1?
(c) C11H12O2? ??IR: 1710 cm?1?
Transcribed Image Text:
TMS 8. 6. O ppm 10 9. 7. Chemical shift (8) TMS O ppm 10 8. 6. Chemical shift (8) TMS O ppm 10 8. 6. Chemical shift (8) Intensity Intensity Intensity 3. 3. 2.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (18 reviews)
a CICHCHCOCH2CH3 b a 138 b 28 ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assign structures to compounds with the following 1H NMR spectra:? (a) C 4 H 7 ClO? ??IR: 1810 cm ?1 ? (b) C 5 H 7 NO 2 ? ?IR: 2250, 1735 cm ?1 ? (c) C 5 H 10 O 2 ? ? ?IR: 1735 cm ?1 ? TMS O ppm 10...
-
Propose structures for compounds X, Y, and Z: The 1H NMR spectrum of X gives two signals, a multiplet at δ 7.3 (5H) and a singlet at δ 4.25 (2H); the 680-840-cm-1 region of...
-
Propose structures for compounds E and F. Compound E (C8H6) reacts with 2 molar equivalents of bromine to form F (C8H6Br4). E has the IR spectrum shown in Fig. 9.50. What are the structures of E and...
-
Chuck, a single taxpayer, earns $86,000 in taxable income and $20,000 in interest from an investment in City of Heflin bonds. (Use the U.S. tax rate schedule.) Required: a. If Chuck earns an...
-
What are primary and secondary data, and why is it best to start your research by gathering secondary data first?
-
A 350-kg roller coaster starts from rest at point A and slides down the frictionless loop-the-loop shown in Fig. P7.45. (a) How fast is this roller coaster moving at point B? (b) How hard does it...
-
Your company has just announced a 7 percent price increase on your entire product line and you are meeting with your most important customer. She announces that your competitor has already been to...
-
Tom Brown, controller of Microbiology Labs, informs you that the company has sold a segment of its business. Mr. Brown also provides you with the following information for 2012: The following...
-
in managing a conflict between two department heads Sam knows that the goals are clearly incompatible which style of conflict is he most likely to choose?
-
Add a new stock, stock 4, to the portfolio optimization model. Assume that the estimated mean and standard deviation of return for stock 4 are 0.125 and 0.175, respectively. Also, assume the...
-
Propose a structure for a compound, C4H7ClO2 that has the following 1R and 1H NMRspectra: 100 80 60 40 20 4000 3000 2500 2000 1000 500 3500 1500 Wavenumber (cm-1) TMS O ppm 10 6. Chemical shift (8)...
-
Epoxy adhesives are prepared in two steps. SN2 reaction of the disodium salt of bisphenol A with epichiorohydrin forms a ?prepolymer,? which is then ?cured? by treatment with a triamine such as H 2...
-
How are intangible assets valued on the balance sheet? Why?
-
Who is generally responsible for providing the accounting department with information n on financing and investment activities n to create the appropriate journal entries in the general ledger?
-
Do we use government wide financial statements or Fund financial statements for financial ratio analysis in government accounting?
-
On October 1 , ABC purchases 1 0 0 units of inventory from XYZ for $ 1 0 0 per unit. These units had a cost of $ 6 0 / unit for XYZ . The terms of the sale are 1 / 1 5 n / 4 5 . On October 5 , ABC...
-
The cash account of Kylie Ltd . Showed a balance of $ 1 6 3 3 5 on December 3 0 , 2 0 2 3 . The bank statement as on that date showed a balance of $ 1 8 , 0 8 6 . After comparing the bank statement...
-
At a level of 7 , 8 5 8 units, Lydia, Inc., a company that makes shovels, has total fixed costs of $ 3 5 9 , 4 3 1 and total variable costs of $ 3 3 6 , 0 5 6 for delivery. What would the total...
-
In the early 1970s, many investors were infatuated by the Nifty 50a group of 50 growth stocks that were thought to be so appealing that they should be bought and never sold, regardless of price....
-
The water in tank A is at 270 F with quality of 10% and mass 1 lbm. It is connected to a piston/cylinder holding constant pressure of 40 psia initially with 1 lbm water at 700 F. The valve is opened,...
-
Name the hybridization scheme that corresponds to each electron geometry. a. Linear b. Trigonal planar c. Tetrahedral d. Trigonal bipyramidal e. Octahedral
-
Which should be faster: bromination of benzene or bromination of N.N-dimethvlaniline? Explain your answer carefully. N,N-dimethylaniline
-
Predict the predominant product(s) from: Mononitration of m-bromoiodobenzene
-
In each of the following sets, rank the compounds in order of increasing harshness of the reaction conditions required to accomplish the indicated reaction. Friedel-Crafts acylation of chlorobenzene,...
-
Question: d. ?During December, Ingrid Legal Services provided legal services, and the client prepaid $7,000. ?Ingrid Legal Services recorded this amount as Unearned Revenue. The job will take several...
-
Question: A company had the following purchases and sales during the month of November:\table[[Date,Activities,Units Acquired at Cost,Units Sold at Retail],[November 1,Beginning inventory,5 units @...
-
Question: Tania, Inc. issued a note payable with principal and interest due at the end of the term of the note. Details about the note payable are as follows:Issuance date: October 1, fear...

Study smarter with the SolutionInn App


