Repeat Exercise 11.22, but with the assumption of chemical equilibrium on stages where catalyst is employed. How
Question:
Repeat Exercise 11.22, but with the assumption of chemical equilibrium on stages where catalyst is employed. How do the results differ from figure?Explain.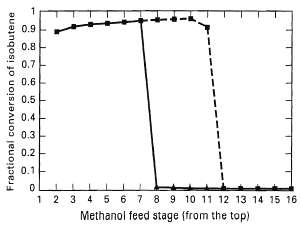
Transcribed Image Text:
0.9 0.8 0,7 0.6 0.5 0.4 0.3 0.2 0.1E 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 Methanol feed stage (from the top) Fractional conversion of isobutene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
The ChemCad program with the SCDS model and reactive distillation option was used to make the equili...View the full answer

Answered By

Arun kumar
made more than four thousand assignments
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Chemical Engineering questions
-
Repeat Example 11.10, but with 10 equilibrium stages instead of 5. What is the effect of this change?
-
Repeat Exercise 11 using Maple with Digits set to 10. In Exercise 11 Use the following values and four-digit rounding arithmetic to construct a third Lagrange polynomial approximation to f (1.09)....
-
Repeat Exercise 6.29, but under the assumption that there are only 20 bearings in the supply bin, 2 of which are defective. In exercise Four wheel bearings are to be replaced on a company vehicle,...
-
The registrar at a prominent northeastern university recently scheduled an instructor to teach two different classes at the same exact time. Help the registrar prevent future mistakes by describing a...
-
Which of the following items is remeasured using the current exchange rate under the temporal method? a. Bonds payable. b. Dividends declared. c. Additional paid-in capital. d. Amortization of...
-
To measure oxygen isotopes in SO24- for geologic studies, SO24- was precipitated with excess Ba2+.24 In the presence of HNO3, BaSO4 precipitate is contaminated by NO3-. The solid can be purified by...
-
A used car dealer sold an automobile to a customer (Robert) on an installment basis. The installment sales contract calls for Robert to make monthly payments of principal and interest. The contract...
-
Refer to Exercise 12- 26. In Exercise 12- 26, Southland Corporation is a small information-systems consulting firm that specializes in helping companies implement standard sales-management software....
-
26 The number of protons, neutrons, electrons in some particles are shown in the table below Particle Protons Neutrons electrons P 1 1 2 Q 2 2 2 R 3 4 2 T 4 5 4 Which one of the following particles...
-
In January 2026, Janeway Inc. doubled the amount of its outstanding stock by selling on the market an additional 10,000 shares to finance an expansion of the business. You propose that this...
-
Repeat Exercise 11.22, but with activities, instead of mole fractions, in the reaction rate expressions. How do the results differ? Explain.
-
An important application of supercritical extraction is the removal of solutes from particles of porous natural materials. Such applications include the extraction of caffeine from coffee beans and...
-
What is the chief complaint?
-
What laws and regulations were broken during the love canal and Hooker chemical incident in New York?
-
Last month McAlister Company had a $60,000 loss on sales of $300,000. Fixed costs are $120,000 a month. Answer the following questions: a. What was the contribution margin percentage? b. What monthly...
-
Why is receptor tyrosine kinase EGFR important for regulation of cell proliferation and cell survival?
-
In class we saw that Karatsuba multiplication allowed one to multiply two n-bit numbers in O(nlog(3)) time. It turns out that using the Fast Fourier Transform, one can multiply numbers in nearly...
-
Kara Ries, Tammy Bax, and Joe Thomas invested $50,000, $66,000, and $74,000, respectively, in a partnership. During its first calendar year, the firm earned $346,200. Prepare the entry to close the...
-
The following nonlinear equations contain terms that are often found in the power flow equations: \[ \begin{aligned} & f_{1}(x)=10 x_{1} \sin x_{2}+2=0 \\ & f_{2}(x)=10\left(x_{1}ight)^{2}-10 x_{1}...
-
APC16550D UART has a clock running at18.432 MHz and its baud rate is set to 2000.Determine the HEX contents of its DLM and DLL registers. Please can you explain step by step and in detail how you get...
-
Ang Electronics, Inc., has developed a new mesh network. If successful, the present value of the payoff (when the product is brought to market) is $29 million. If the mesh network fails, the present...
-
A filtration system for a fermentation effluent is being designed on the basis of the data taken using a pilot-plant plate-and-frame filter press. The filter area is 0.3 m 2 , cF = 2,000 kg/m 3 , P =...
-
A test leaf filter is used to determine the filtration rate for an unclarified broth from a fermentor. At a pressure drop of 50 kPa, 150 mL of filtrate are collected in 30 minutes. If the resistance...
-
In a filtration cycle, why does constant-pressure filtration usually occur near the end of the cycle and constant-rate filtration at the beginning?
-
In the Queries section of the Navigation Pane, right-click Stone Mountain Patients to select it and display the shortcut menu. 3. Click Copy on the shortcut menu. 4. Right-click the empty area near...
-
What is 'audit risk', and discuss/analyze the components of audit risk. b. List the four (4) things involved in the initial audit planning and which should be done early in the audit.
-
K Internal control is a plan of organization and a system of procedures, implemented by company. A. external auditors; management B. external auditors; board of directors OC. internal auditors;...

Study smarter with the SolutionInn App


