Sketch the atomic packing of (a) The (100) plane for the BCC crystal structure, and (b) The
Question:
Sketch the atomic packing of
(a) The (100) plane for the BCC crystal structure, and
(b) The (201) plane for the FCC crystal structure (similar to Figures 3.10b and3.11b).
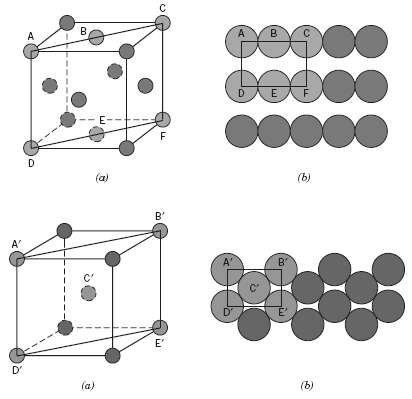
Transcribed Image Text:
00000 E- (a) (b) B' A' A' B" D' (b) (a)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (16 reviews)
a A BCC unit cell its 100 plane and the atomic packing of this plane are ind...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
One slip system for the BCC crystal structure is {110}. In a manner similar to Figure, sketch a {110}-type plane for the BCC structure, representing atom positions with circles. Now, using arrows,...
-
One slip system for the BCC crystal structure is {110}(111). In a manner similar to Figure 7.6b, sketch a {110}-type plane for the BCC structure, representing atom positions with circles. Now, using...
-
For both FCC and BCC crystal structures, there are two different types of interstitial sites. In each case, one site is larger than the other, and is normally occupied by impurity atoms. For FCC,...
-
You name the catastrophe, and JIT has been through it and survived. Toyota Motor Corporation has had its world-renowned JIT system tested by fire. The massive fire incinerated the main source of...
-
What happens to the air pressure inside a balloon when it is squeezed to half its volume at constant temperature?
-
Which of these is seen in Asherman syndrome? a) Oligomenorrhea b) Hypomenorrhea c) Metromenorrhagia d) Polymenorrhea
-
You are calibrating magnetic coils for a particle detector. One step involves checking the magnetic field at different positions inside a toroid, and you are asked to measure the field at the...
-
The plaintiff, Herbert Rosenthal Jewelry Corporation, and the defendant, Kalpakian, manufactured jewelry. The plaintiff obtained a copyright registration of a jeweled pin in the shape of a bee....
-
1 Explain briefly about six sigma and capacity maturity models? 2 Explain about the contemporary management practices taking place in Indian business model? 3 4 What is performance management?...
-
Explain the difference between closed-fact and open-fact situations.
-
Cite the indices of the direction that results from the intersection of each of the following pair of planes within a cubic crystal: (a) (100) and (010) planes, (b) (111) and (111) planes, and (c)...
-
Consider the reduced-sphere unit cell shown in Problem 3.20, having an origin of the coordinate system positioned at the atom labeled with an O. For the following sets of planes, determine which are...
-
1. What are the coordinates of the point Q in Fig. 7? 2. What are the coordinates of the point P in Fig. 7?
-
How can index futures be used to change the beta of a well-diversified portfolio?
-
Explain how LIBOR is determined and why it is being discontinued.
-
How is the formula for the minimum variance hedge ratio changed to take account of daily settlement?
-
Explain what liquidity preference theory and expectations theory imply about forward interest rates and expected future spot interest rates.
-
What is the relationship between the quoted price and cash price of a Treasury bill?
-
The Greenpeace Report issued in 2020, UNPACKED: How supermarkets can cut plastic packaging in half by 2025, suggested that: GOVERNMENT SHOULD: Set legally binding targets in the Environment Bill to...
-
Explain the operation of the dividends received deduction.
-
Sketch the region under the graph of the function and find its area using FTC I. f(x)=2x-x, [0,2]
-
A cylindrical specimen of a brass alloy 7.5 mm (0.30 in.) in diameter and 90.0 mm (3.54 in.) long is pulled in tension with a force of 6000 N (1350 lbf); the force is subsequently released. (a)...
-
A steel bar 100 mm (4.0 in.) long and having a square cross section 20 mm (0.8 in.) on an edge is pulled in tension with a load of 89,000 N (20,000 lbf), and experiences an elongation of 0.10 mm (4.0...
-
A steel alloy specimen having a rectangular cross section of dimensions 12.7 mm 6.4 mm (0.5 in. 0.25 in.) has the stress-strain behavior shown in Figure 6.21. If this specimen is subjected to a...
-
If you were running a campaign that had lower than expected impressions, what should you do?
-
1. Develop a definition for the Triple C model of project management. 2. List some of the factors that can impede the flow of information for project planning purposes. How can these factors be...
-
The Meat Mart has $900,000 in net income. The firm has 200,000 shares of stock outstanding. The market price per share is $76. What is the PE (price to earnings) ratio?

Study smarter with the SolutionInn App


