Figure 9.27 shows a duct in which methyl alcohol at 25°C flows at the rate of 3000
Question:
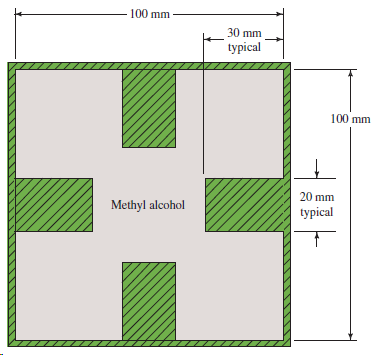
Transcribed Image Text:
100 mm 30 mm typical 100 mm 20 mm Methyl alcohol typical
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
A0100 m 4002003m WP40108003064 m Q3000 L...View the full answer

Answered By

Chiranjib Thakur
I have no tutoring experience yet, but I can share my skills and knowledge gained from my education and work experiences. I have been a CPA since 2012 with 6 years of work experience in internal auditing and 4 years of work experience in accounting at the supervisory level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A hair dryer is basically a duct in which a few layers of electric resistors are placed. A small fan pulls the air in and forces it through the resistors where it is heated. Air enters a 1200-W hair...
-
Consider conditions for which a mercury-in-glass thermometer of 4-mm diameter is inserted to a length L through the wall of a duct in which air at 77C is flowing. If the stem of the thermometer at...
-
Figure 9.29 shows a system in which methyl alcohol at 77°F flows outside the three tubes while ethyl alcohol at 0°F flows inside the tubes. Compute the volume flow rate of each fluid required...
-
The table shows equivalent temperatures in degrees Celsius and degrees Fahrenheit. (a) Plot the data with Fahrenheit temperature on the x-axis and Celsius temperature on the y-axis. What type of...
-
On June 1, 2014, Novack Company purchases equipment on account from Moleski Manufacturers for $50,000. Novack is unable to pay its account on July 1, 2014, so Moleski agrees to accept a three-month,...
-
11. What is the difference between the below two variables? a) $NewEmployee = "Sammy" b) $EmployeeNames = "CN=MyBoss","CN=MyDirect1","CN=MyDirect2"
-
Consider the delivery time data discussed in Example 11.3. Find the PRESS statistic for the model developed from the estimation data. How well is the model likely to perform as a predictor? Compare...
-
The 0.5-kg flyballs of a centrifugal governor revolve at a constant speed v in the horizontal circle of 150-mm radius shown. Neglecting the mass of links AB, BC, AD, and DE and requiring that the...
-
Audit reporting processses inclusive of "what the report would be against and to whom the report may be accessible to by what methods" and rmore clarification about audit reporting ?
-
36. Star Adventures has invested financial and time resources into ________ because they want their employees to develop teamwork skills, using challenging and structured activities. simulations...
-
A furnace heat exchanger has a cross section like that shown in Fig. 9.28. The air flows around the three thin passages in which hot gases flow. The air is at 140°F and has a density of 2.06...
-
In Fig. 9.26, ethylene glycol (sg = 1.10) at 77°F flows around the tubes and inside the rectangular passage. Calculate the volume flow rate of ethylene glycol in gal/min required for the flow to...
-
Using the Bohr model, estimate the wavelength of the Ka X-ray in nickel (Z = 28).
-
Convert the context-free grammar G in Chomsky Normal Form, where G = (V,T, S,T) with V = {S, A, B, C, D), T = {a,b,c} and P: S AABD|aBB A a| B BB|bc|X CCD|a D Db DD
-
Consider the following cash flows and interest rates: 0 r = 2.5% 1 r = 1.5% 5,000 2 r = 1.9% 5,000 3 r = 2.4% 5,000 4 5,000 a. Calculate the present value of these cash flows (at time 0). (5 marks)...
-
How can legislation and regulation impact on OD and change?
-
The purpose of this assignment is to write a program that uses loops, file I/O, and methods to process employee salary data and generate a small report. Additionally, you will gain further practice...
-
List some of the indications or clues that may arise due to conflict in the workplace? and why
-
Identify and discuss appropriate modes of transportation for the following items: a. Apple iPhones b. Under Armour running shoes c. Organic fruits and vegetables d. Pressure treated lumber
-
[a] Two foam blocks, each with a charge of 19 micro coulombs (1 C = 10-6 C), are both held in place 19 cm apart in the east-west direction. A foam ball with a charge 49 C is placed 55 cm north of the...
-
A sample containing 42.1 g of Ar is enclosed in a container of volume 0.0885 L at 375 K. Calculate P using the ideal gas, van der Waals, and RedlichKwong equations of state. Based on your results,...
-
The experimental critical constants of CH 4 are found in Table 7.2. Use the values of Pc and T c to calculate V c . Assume that CH 4 behaves as a. An ideal gas b. A van der Waals gas c. A...
-
The observed Boyle temperatures of H 2 , N 2 , and CH4 are 110, 327, and 510. K, respectively. Compare these values with those calculated for a van der Waals gas with the appropriate parameters.
-
A project that is expected to generate annual after-tax cash flows of $2,200 for each of the next 20 years. The project's cost of capital is 8.5 percent and it has an IRR of 9.058 percent. What is...
-
Gabbie has to decide between the following two choices: Option 1: Get $320 with probability 35.97%, and $84.15 with the complement probability. Option 2: Get $94.95 with 100% probability. Her utility...
-
Why are unpaid accruals a "temporary" source of cash? Please give some details and examples.

Study smarter with the SolutionInn App


