0.10 mol of gas undergoes the process 1 2 shown in Figure P 16.59. a. What...
Question:
0.10 mol of gas undergoes the process 1 → 2 shown in Figure P 16.59.
a. What are temperatures T1 and T2 (in °C)?
b. What type of process is this?
c. The gas undergoes an isothermal compression from point 2 until the volume is restored to the value it had at point 1. What is the final pressure of the gas?
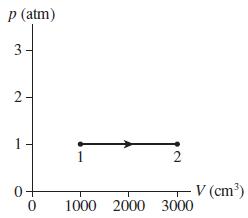
Transcribed Image Text:
p (atm) 3- 1 V (cm³) 1000 2000 3000 2. 2.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
Model Assume the gas is an ideal gas Solve a We can find the temperatures di...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Physics For Scientists And Engineers A Strategic Approach With Modern Physics
ISBN: 9780321740908
3rd Edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Mathematics questions
-
0.0050 mol of gas undergoes the process 1?? 2 ?? 3 shown in Figure P16.38.? What are (a) Temperature T 1 , (b) Pressure p 2 , (c) Volume V 3 ? p (atm) P2 - T = 2926 K 3 T3 = 2438 K 1 0+ V (cm) V3...
-
0.0040 mol of gas undergoes the process shown in FIGURE EX18.35. a. What type of process is this? b. What are the initial and final temperatures in °C? p (atm) 3- 2- V (cm) 300 0+ 100 200 FIGURE...
-
0.020 mol of gas undergoes the process shown in FIGURE EX18.37. a. What type of process is this? b. What is the final temperature in °C? c. What is the final volume V 2 ? p (atm) 3- 2- 1- V (cm)...
-
Given are five observations for two variables, x and y. a. Develop a scatter diagram for these data. b. What does the scatter diagram developed in part (a) indicate about the relationship between the...
-
In the production of ethanol, the feedstock high in sugar content is first converted to sugar, and the sugar (glucose) is fermented into ethanol through the reaction C6H12O6 2 C2H5OH + 2 CO2....
-
Perform the indicated operations. 9x + 46x + 5 3x2x1 + x + 11x + 30 x + 5x - 6x
-
A homeowner found a preferred savings account book in the wall of a home he was remodelling. He went to the bank and discovered that the account was 25 years old and had $\$ 425,801$ in the bank! The...
-
Hartwell Drug Company produces a supplement to improve bone density. Conversion costs are added evenly throughout the production process. The following information is available for March: Required a....
-
Sodium chloride is the primary salt dissolved in ocean water. For every liter, there are about 35 grams of salt dissolved in ocean water, which corresponds to a salt concentration of 0.6 moles/liter,...
-
Lots of data are available to retailers to make good decisionsloyalty programs, Web analytics, and point-of-sale data. However, there is a big gap between having data and being able to leverage them...
-
A gas cylinder with a tight-fitting, movable piston contains 200 cm 3 of air at 1.0 atm. It floats on the surface of a swimming pool filled with 15C water. The cylinder is then pulled slowly...
-
Compare and contrast software engineering with other engineering disciplines.
-
Ajax Limited reported the following information (in millions) at December 31, 2018: net sales $14,000; net income $550; total assets at December 31, 2018, $7,200; and total assets at December 31,...
-
Explain the concept of adaptable organization and state an example of both, organizations that proved to be adaptable as well as of those that failed to transform and adapt to their environment....
-
Determine the amount of tax liability in the following situations. In all cases, the taxpayer is using the filing status of married filing jointly. Use the Tax Tables for taxpayers with taxable...
-
How do authors employ intertextual allusions to enrich the layers of meaning in their works and establish connections across disparate literary traditions?
-
A company's inventory records indicate the following data for the month of April: Date Activities Units Acquired at Cost Units Sold at Retail April 1 Beginning inventory 8 2 0 units @ $ 3 6 = $ 2 9 ,...
-
The Pallon Company manufactures a variety of handerated bed frames. The company's manufacturing activitiesor the current Sear follow:Manufacturing Activity Estimated Cost Cost Driver Used as...
-
Street, Rhode and Close carried on business in partnership sharing profits and losses, in the ratio 5 : 4 : 3. Their draft statement of financial position as on 31 March 20X2 was as follows: Street...
-
Copy and complete the statement. 3800 m ? km =
-
Hookes law describes an ideal spring. Many real springs are better described by the restoring force (F Sp ) s = -ks - q(s) 3 , where q is a constant. Consider a spring with k = 250 N/m and q = 800...
-
Hookes law describes an ideal spring. Many real springs are better described by the restoring force (F Sp ) s = -ks - q(s) 3 , where q is a constant. Consider a spring with k = 250 N/m and q = 800...
-
The force acting on a particle is F x = F 0 e-x/L . How much work does this force do as the particle moves along the x-axis from x = 0 to x = L?
-
Gilbert Canned Produce (GCP) packs and sells three varieties of canned produce: green beans; sweet peas; and tomatoes. The company is currently operating at 82 percent of capacity. Worried about the...
-
The accounting records of Allen Insulation, Inc. reflected the following balances as of January 1, 20x0: Beginning Inventory: $190 units @ $120 The following transactions occurred in 20x0: January...
-
Sarasota Limited has a calendar-year accounting period. The following errors were discovered in 2023. 1. The December 31, 2021 merchandise inventory had been understated by $50,400. 2. 3. Merchandise...

Study smarter with the SolutionInn App


