In Exercises find (a) ( o g) (-1) (b) (g o ) (2) (c) ( o )
Question:
In Exercises find
(a) (∫ o g) (-1)
(b) (g o ∫) (2)
(c) (∫ o ∫) (x)
(d) (g o g) (x)
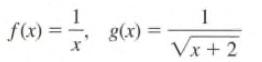
Transcribed Image Text:
f(x) = — g(x) = 1 Vx+2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
fx 1x gx ...View the full answer

Answered By

Hardik Dudhat
I am semi-qualified Chemical Engineering ,I have scored centum in accounting in my senior secondary and in my graduation. I have always helped my fellow students with their concerns on the subject, i have tutored on various tutoring sites in the past and also have taken home tuitions for degree and MBA students. As a tutor, I don't want my students to just get a solution, I want them to understand the concept and never have a doubt in that area thereon and i believe in excelling and not in educating.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Calculus Graphical, Numerical, Algebraic
ISBN: 9780132014083
3rd Edition
Authors: Ross L. Finney, Franklin D. Demana, Bert K. Waits, Daniel Kennedy
Question Posted:
Students also viewed these Mathematics questions
-
Starting from any of the monomers given above (A, B, C or D), propose a synthesis of a responsive polymer capable of sensing Nat ions, detail the proposed response and how this could be observed...
-
In Exercises find a parametrization for the curve. The line segment with endpoints (-1, -3) and (4, 1)
-
In Exercises find a parametrization for the curve. The line segment with endpoints (-1, 3) and (3, -2)
-
What is SAV? What are some economic forces that can help explain SAV? What are some demographic and other considerations? How might physician uncertainty lead to SAV?
-
In an investigation of environmental causes of disease, data were collected on the annual mortality rate (deaths per 100,000) for males in 61 large towns in England and Wales. In addition, the water...
-
Brandon Company produces and sells a product that has variable costs of $8 per unit and fixed costs of $250,000 per year. 1. Compute the unit cost at a production and sales level of 10,000 units per...
-
Differentiate technology from methodology and from method. Can you come up with an example that differentiates these concepts in a specific context, perhaps software development?
-
Assume the same facts as for Question 1 above. The fair value of the investment in Company E is $220,000 on December 31, 2011. Answer the following questions assuming the investment is recorded using...
-
Correlle dishes are known for being mostly unbreakable in daily use. Kevin's 12-year-old son decided to test this "unbreakable" idea by throwing a plate hard onto the floor. It broke. what this is an...
-
How is trading-area analysis different for a small store than a large store? Explain your answer. How should a food-delivery service such as Amazon Fresh define its trading area? Is it going to...
-
In Exercises evaluate the expression in (cos (77)) 11
-
Which of the following describes the graph of the parametric curve x = - 3 sin t, y = - 3 cos t? (A) Circle (B) Parabola (C) Ellipse (D) Hyperbola (E) Line
-
What are the necessary conditions for economic efficiency? In what four situations might a market fail to achieve ideal economic efficiency?
-
A \(\frac{1}{3}\)-mol sample of an ideal gas is sealed in a container and heated from \(273 \mathrm{~K}\) to \(500 \mathrm{~K}\). For this gas, what is the value of \(C_{P}\) in units of \(k_{B}\) if...
-
An ideal gas for which \(\gamma=1.4\) initially has a volume of \(1.5 \mathrm{~m}^{3}\) and a pressure of \(15 \mathrm{MPa}\). The gas is then slowly compressed isentropically to a volume of \(0.50...
-
It is somerimes said, erroneously, that biological organisms violate the second law of thermodynamics because the biochemical reactions they carry out reduce their entropy by converting incoherent...
-
A 22.7-g sample of diatomic oxygen gas molecules is in a sealed rigid container that has a volume of \(0.0240 \mathrm{~m}^{3}\). If the gas is initially at \(-1.50^{\circ} \mathrm{C}\), by how much...
-
An ideal gas undergoes an isentropic process that changes its pressure from \(P_{i}\) to \(P_{f}\) and its volume from \(V_{\mathrm{i}}\) to \(V_{t}\). What is the work done on the gas?
-
True or false? (a) The reduced mass of a two-particle system is always less than m1 and less than m2. (b) When we solve a two-particle system (whose potential-energy V is a function of only the...
-
Show that the block upper triangular matrix A in Example 5 is invertible if and only if both A 11 and A 22 are invertible. Data from in Example 5 EXAMPLE 5 A matrix of the form A = [ A11 A12 0 A22 is...
-
The study reported 45 injuries to the upper extremity on a dry field consisting of artificial turf over the course of 10,112 games. What procedure can be used to assess whether the risk of injury is...
-
Provide a 95% CI for the rate of injury to the upper extremity on artificial turf. Express each rate as the number of injuries per 1000 games? Sports Medicine Injuries are common in football and may...
-
Her doctor is considering taking her off antihypertensive medications but wants to be fairly certain that her true DBP is less than 90 mm Hg. Use a statistical approach to answer this question....
-
Large-scale industrial production - accompanied by massive technological change, expanding international communication networks, and pro-growth government policies-generated rapid economic...
-
How can this relate to individuals who have had a TBI and are currently homeless or marginally housed?
-
1. According to Marxian theory, are crises an economic process inherent to the accumulation dynamics of current capitalism? Justify your answer. 2. According to Marxian theory, what do you consider...

Study smarter with the SolutionInn App


