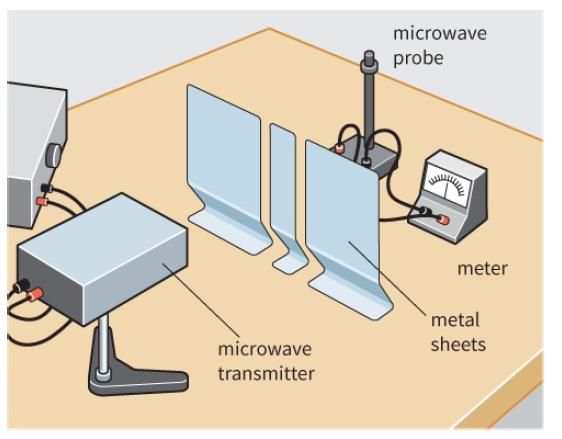
Look at the experimental arrangement shown in Figure 13.17. Suppose that the microwave probe is placed at
Question:
Look at the experimental arrangement shown in Figure 13.17. Suppose that the microwave probe is placed at a point of low intensity in the interference pattern.
Suggest what will happen if one of the gaps in the barrier is now blocked.
Transcribed Image Text:
microwave probe meter metal microwave sheets transmitter
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
Blocking one of the gaps in the barrier will result in a change in the interference pattern ca...View the full answer

Answered By

Akshay Shete
I have extensive experience as a tutor, both online and in-person. I have worked with students of all ages and abilities, and am skilled at adapting my teaching style to meet the needs of each individual student. I have a strong background in a variety of subjects, including math, science, and English, and am able to break down complex concepts in a way that is easy for students to understand. In addition to my subject matter expertise, I am also a patient and supportive teacher, and am committed to helping my students succeed. Whether I am working with a struggling student who needs extra help to catch up, or an advanced student looking to get ahead, I am able to provide the guidance and support they need to reach their goals. Overall, my hands-on experience as a tutor has prepared me to be a confident and effective teacher, and I am excited to use my skills to help students succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Physics Coursebook
ISBN: 9781108859035
3rd Edition
Authors: David Sang, Graham Jones, Gurinder Chadha, Richard Woodside
Question Posted:
Students also viewed these Sciences questions
-
In the apparatus shown below, what will happen if the membrane is (a) Permeable to both water and the Na+ and Cl- ions, (b) Permeable to water and Na+ ions but not to Cl- ions, (c) Permeable to water...
-
In the arrangement shown in Figure P18.21, an object can be hung from a string (with linear mass density μ = 0.002 00 kg/m) that passes over a light pulley. The string is connected to a vibrator...
-
In real-time interactive audio/video, what will happen if a packet arrives at the receiver site after the scheduled playback time?
-
Determine the beam column shown below is capable of safely carrying the applied loads. Given loads were determined by using LRFD provisions and bending moment diagrams are obtained from second order...
-
The owner of the Blue Goose Motel borrowed $12 500 at 7.1% compounded semiannually and agreed to repay the loan by making payments of $700 at the end of every three months. (a) How many payments will...
-
Kidman has just completed a review of its job-costing system. This review included a detailed analysis of how past jobs used the firms resources and interviews with personnel about what factors drive...
-
Table 1.2 illustrates a possible scenario in a trade on gold futures. On day 1 , when the gold futures price is \(\$ 1350\) per ounce, we enter a long position for two contracts, whose unit size is...
-
"The transactions completed by Revere Courier Company during December, the first month of the fiscal year, were as follows: Dec. 1. Issued Check No. 610 for December rent, $4,200. 2. Issued Invoice...
-
Draw energy band diagram for a MOS structure in thermal equilibrium with (a). P-type semiconductor. Use4 , 3.5 , 1.0m sc Gq eV q eV E eV = = = and calculate VFB. Estimate the required applied voltage...
-
The frequency of a tuning fork is 505 Hz. Find the length of the shortest organ pipe which will give 6 beats/s with the fork. The velocity of sound in air is 336.5m/s.
-
Explain why, in remote mountainous regions, such as the Hindu Kush, radio signals from terrestrial transmitters can be received, but television reception can only be received from satellite...
-
State how the diffracted pattern will change when: a. The wavelength of the incident wave is increased b. The wavelength of the incident wave is decreased.
-
Record the transactions in E6A in the general journal. In E6A a. Collin Ferdinand opened Ferdinand Repair Service, Inc., by investing $8,600 in cash and $3,200 in repair equipment in return for...
-
Write the object code for each statement in the following SIC / S program and create an object code file. In other words, describe the actual address of each statement and the assembled object code,...
-
You can buy EURUSD in the London market at 1.1900, you can buy USDCHF in New York at 0.9500, and you can sell EURCHF in Zurich at 1.1405. Given this information, is there an arbitrage opportunity?...
-
You will be provided a car price of either: $12k, $16k, $20k, $24k, $28k, $32k, $36k, $40k, $44k, or $48k 1. Suppose you have $4000 saved up for a down payment, and you plan to finance the rest of...
-
Naiser has current wages of $ 1 5 , 6 0 0 . 0 0 and year - to - date earnings of $ 2 0 8 , 0 0 0 . Naiser has 2 0 2 2 HI withholding, for this pay period, is: $ The supplemental withholding, for this...
-
Medicare has seen a steady increase and reliance on non-physician practitioners (such as Nurse Practitioners and Physician Assistants) to treat patients. Explain how Medicare reimburses for their...
-
An ideal solenoid has length . If the windings are compressed so that the length of the solenoid is reduced to 0.50, what happens to the inductance of the solenoid?
-
The diagram shows the two forces acting on a small object. Which of the following is the resultant force on the object? A. 8 N downwards B. 8 N upwards C. 2 N downwards D. 2 N upwards 3 N 5 N
-
If the wall depicted in Figure 1.2 were a movable piston, under what conditions would it move as a result of the molecular collisions? Figure 1.2 mvx mvx
-
The mass of a He atom is less than that of an Ar atom. Does that mean that because of its larger mass, Argon exerts a higher pressure on the container walls than He at the same molar density, volume,...
-
Identify the reagents necessary to achieve each transformation below. In each case, you will need to use at least one reaction from this chapter and at least one reaction from the previous chapter....
-
A metal sample weighing 71.9 g and at temperature of 100.0 degrees celsius was placed in 41.0g of water in a calorimeter at 24.5 degrees celsius. At equilibrium, the temperature of the water and...
-
Match each leadership theory with a corresponding related management approach: leadership theories: situational leadership, transformational leadership, Theory "Y", Theory "X" management approach:...
-
An important practice is to check the validity of any data set that you analyze. One goal is to detect typos in the data, and another would be to detect faulty measurements. Recall that outliers are...

Study smarter with the SolutionInn App


