Look at Figure 29.27. a. What is the retention time of the compound shown? b. What is
Question:
Look at Figure 29.27.
a. What is the retention time of the compound shown?
b. What is the approximate relative molecular mass of the compound shown?
c. How would the compound be identified?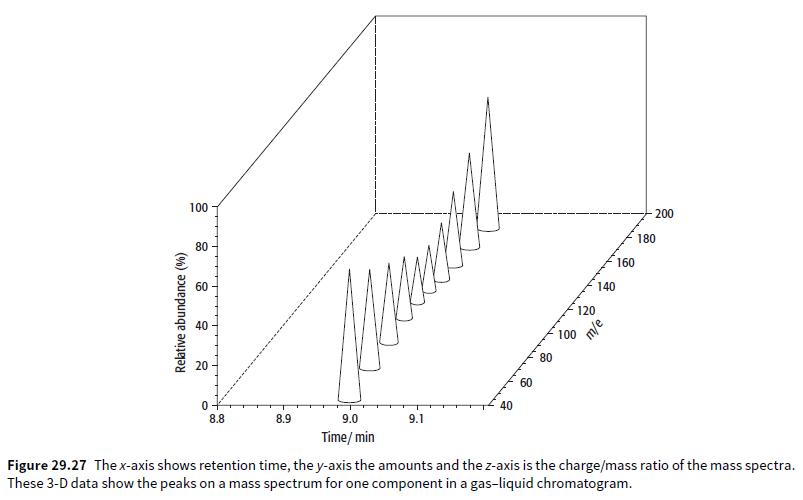
Transcribed Image Text:
100 200 80 180 160 60 140 120 40 100 20 80 60 40 8.8 8.9 9.0 9.1 Time/ min Figure 29.27 Thex-axis shows retention time, the y-axis the amounts and the z-axis is the charge/mass ratio of the mass spectra. These 3-D data show the peaks on a mass spectrum for one component in a gas-liquid chromatogram. Relative abundance (%)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
a We have to answer what is the retention time of the compound shown The retention time ...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Students also viewed these Sciences questions
-
Use this information to compute the following: FunTime Cruiseline offers nightly dinner cruises departing from several cities on the eastern coast of the United States including Charleston,...
-
A large distributor of oil-well drilling equipment operated over the past two years with EOQ policies based on an annual holding cost rate of 28%. Under the EOQ policy, a particular product has been...
-
(a) Make a graph showing the retention time of each peak in Figure 24-25 in chromatograms B, F, and C as a function of position along the line BC. Predict the retention times for solvent compositions...
-
Swish Designs specialises in designing commercial office space in Auckland. The CEO, Ralph Polo has reviewed the financial results and has noticed that operating profits were below budget. He also...
-
The owner of a sporting goods store is considering remodeling the store to carry a larger inventory. The cost of remodeling and additional inventory is $60 000. The expected increase in net profit is...
-
From the following, calculate departmental income before tax. Assume a tax rate of30%. Dept. A Dept. B Net Sales Cost of Goods Sold Detivery Expense Advertising Expense Depreciation Expense $210,000...
-
Wire \(1,5.0 \mathrm{~m}\) long and carrying a current of \(3.0 \mathrm{~A}\), experiences a magnetic force of magnitude \(4.0 \times 10^{-7} \mathrm{~N}\) when placed \(90 \mathrm{~mm}\) away from...
-
Kegler Company has set the following standard costs per unit for the product it manufactures. Direct materials (15 Ibs. @ $ 4 per Ib.) . . . . . . . . . . . $ 60.00 Direct labor (3 hrs. @ $ 15 per...
-
(a) Equity of KGF Ltd. (KGFL) is Rs. 410 Crores, its debt, is worth Rs. 170 Crores. Printer Division segments value is attributable to 74%, which has an Asset Beta (p) of 1.45, balance value is...
-
You are part of the engagement team for the audit of Suzuki Manufacturing for the year ended December 31, 2019, and are responsible for auditing the acquisition cycle. Download the Excel file for the...
-
Look at Figure 29.28. a. Calculate the relative molecular mass of leucine enkephalin (C 28 H 37 N 5 O 7 ) using relative atomic masses. (A r values C = 12.0, H = 1.0, N = 14.0, O = 16.0) b. i. How is...
-
a. List the ions responsible for the M, [M + 2] and [M + 4] peaks in a mass spectrum of dibromomethane. b. What would be the mass-to-charge ratio and relative abundances of the major peaks with the...
-
Evaluate the following integrals. S." 2sin x cos x dx
-
1. The new public management is at the origin of the transformations of the public administration and consequently, of the health system. Present and describe two contributions of public management...
-
Understand the importance of leadership for quality Describe the role of leaders in performance excellence Apply examples of leaders who have inspired their organizations to achieve quality and...
-
A student performs the following lead extraction to test the stoichiometric method and to determine the efficiency of the reaction. In the reaction49.5 g of lead (II) nitrateis reacted with excess zi...
-
A wave has a speed of 1200 m/s and a wavelength of 3.3 m. What is its approximate frequency?
-
Evaluate the advantages and disadvantages of both symmetrical and master-slave multiprocessing systems in regards to computer processing speed, multiprocessing configuration, overheating, and cost....
-
Light of wavelength 692 nm in air passes into window glass with an index of refraction of 1.52 (a) What is the wavelength of the light inside the glass? (b) What is the frequency of the light inside...
-
A researcher reports a significant two-way between-subjects ANOVA, F(3, 40) = 2.96. State the decision to retain or reject the null hypothesis for this test.
-
For each of the following pairs of compounds, identify the higher boiling compound and justify your choice: a. b.
-
Refer to Figure 1.10 and explain why (U/V) T is generally small for a real gas. Figure 1.10 Ideal gas Real gas Tranlaition rv-0 (4)A
-
Can a gas be liquefied through an isenthalpic expansion if J T = 0?
-
xPlease address the questions below in a memorandum to Apex Investment Partners. Please include your revised capitalization tables and waterfall diagrams. How, if at all, would you change the...
-
Sky Company employed Tom Mills in Year 1. Tom earned $5,700 per month and worked the entire year. Assume the Social Security tax rate is 6 percent for the first $110,000 of earnings, and the Medicare...
-
Pioli Corporation manufactures one product. It does not maintain any beginning or ending Work in Process inventories. The company uses a standard cost system in which inventories are recorded at...

Study smarter with the SolutionInn App


