Look at the mass spectrum of germanium, Ge. a. Write the isotopic formula for the heaviest isotope
Question:
Look at the mass spectrum of germanium, Ge.
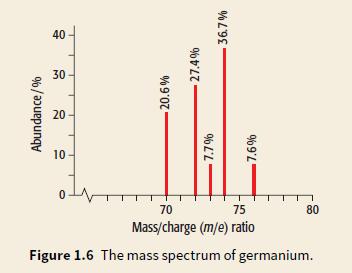
a. Write the isotopic formula for the heaviest isotope of germanium.
b. Use the % abundance of each isotope to calculate the relative atomic mass of germanium.
Transcribed Image Text:
40 30 20 10 70 75 80 Mass/charge (m/e) ratio Figure 1.6 The mass spectrum of germanium. Abundance/% 20.6% 27.4% 96 L'L 36.7%
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
a According to the mass spectrum of germanium Ge provided in the question the isotopic formul...View the full answer

Answered By

SHAKIL AHMED
I worked as a homework/assignment helper for more than 4 years. I also worked as a teaching assistant for Bachelor's students. I loved teaching them. Interaction with the students help me to grow as a teacher and I learned a lot while teaching them.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Students also viewed these Sciences questions
-
The mass spectrum of methyl isobutyl ether does not show a peak due to inductive cleavage, in contrast to the mass spectrum of di-sec-butyl ether (Eq. 12.31). Use what you know about carbocation...
-
The mass spectrum of 1-butanol shows an intense daughter ion peak at m/z 5 31. Explain how this peak might arise.
-
The mass spectrum of n-octane shows a prominent molecular ion peak (m z 114). There is also a large peak at m/z 57, but it is not the base peak. The mass spectrum of 3,4-dimethylhexane shows a...
-
Timco is considering the construction of a new retail outlet. The construction cost will be 400000. Net working capital will increase by 10000. The depreciation is 10 year MACRS. The new location...
-
Give atomic symbols for each element. a. Sodium b. Argon c. Nitrogen d. Radon
-
Since its inception, Inman Laboratory has produced a single product, Product S109. With the advent of automation, the company added the technological capability to begin producing a second product,...
-
In Figure P27.62, an external magnetic field is directed out of the page, and six wires are placed, one at a time, in this field. Each wire carries a current in the direction indicated; if there is...
-
Mighty Stars produces stars for elementary teachers to reward their students. Mighty Stars' trial balance on June 1 follows: MIGHTY STARS Trial Balance June 1, 2018 June 1 balances in the subsidiary...
-
Write the SOP Boolean expressions for the 7 outputs. [ 3 . 5 marks ]
-
Maximum Pension Fund is attempting to manage one of the bond portfolios under its management. The fund has identified three bonds which have five year maturities and trade at a yield to maturity of 9...
-
Solid sodium carbonate reacts with aqueous hydrochloric acid to form aqueous sodium chloride, carbon dioxide and water. Na 2 CO 3 + 2HCl 2NaCl + CO 2 + H 2 O a. Rewrite this equation to include...
-
This question is about two transition metals, hafnium (Hf) and zirconium (Zr). a. Hafnium forms a peroxide whose formula can be written as HfO 3 .2H 2 O. Use the A r values below to calculate the...
-
Why is an explicit transaction end statement needed in SQL but not an explicit begin statement?
-
Why use a PivotTable? What is the main function of a PivotTable? Are data and information the same thing? What is the difference between raw data and information? Is information in a spreadsheet...
-
Bank A in Mexico City posts a letter of credit with an American company as the seller-beneficiary in the transaction. How will A typically advise the seller that the letter of credit is available for...
-
A big tank initially contains 1000 L of a salt-water solution, which has a concentration of 0.02 kg of salt per liter. Brine (a solution of salt in water) containing 0.03 kg of salt per liter of...
-
We are all watching the Super Bowl this Sunday. Let's say Eagle Birdie said to Chief Kanssy "I bet you 1000.00 that the Philadelphia Eagles will win BY TEN POINTS in the Super bowl" Chief Kanssy...
-
Explain the following terms: Consent Implied and expressed consent Consent to medical treatment Self Defence Provocation Necessity Duress Legal Authority Part 11 what is meant by a true defence? what...
-
a. Give the major product of each of the following reactions: 1. 2. 3. 4. b. What do all the reactions have in common? c. How do all the reactions differ? CH CH Ht CH Ht CH3C-CH2
-
Don Griffin worked as an accountant at a local accounting firm for five years after graduating from university. Recently, he opened his own accounting practice, which he operates as a corporation....
-
Identify the reagents you would use to achieve each of the following transformations: a) Convert tert-butyl bromide into a primary alkyl halide b) Convert 2-bromopropane into 1-bromopropane
-
Identify the reagents necessary to accomplish each of the transformations shown below. If you are having trouble, the reagents for these transformations appear on page 444, but you should first try...
-
Identify the reagents you would use to accomplish each of the following transformations: (a) Convert 2-methyl-2-butene into a monosubstituted alkene (b) Convert 2,3-dimethyl-1-hexene into a...
-
Construction Manager Certification Institute What types of certifications are offered? Are these certifications required or voluntary? What are some advantages to having these certifications? What is...
-
The challenges involved in managing virtual teams. As a new manager, you are tasked with overseeing a team that has been functioning poorly for over a year. What is one team strategy that you decide...
-
According to Scholar Partitional Leader How do you plan to model SPL daily within the school and community?

Study smarter with the SolutionInn App


