(a) Determine the charge of the aluminosilicate ion whose composition is AlSi 3 O 10 . (b)...
Question:
(a) Determine the charge of the aluminosilicate ion whose composition is AlSi3O10.
(b) Using Figure 22.32, propose a reasonable description of the structure of this aluminosilicate.
Figure 22.32
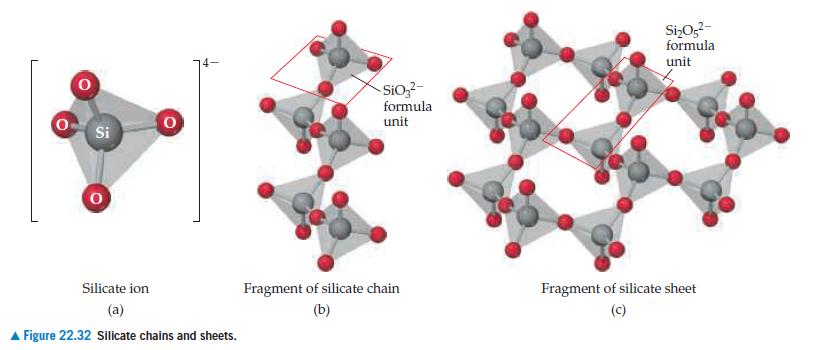
Transcribed Image Text:
Si 0 Silicate ion (a) ▲ Figure 22.32 Silicate chains and sheets. SiO3²- formula unit Fragment of silicate chain (b) Si₂05²- formula unit Fragment of silicate sheet (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
a The charge of the aluminosilicate ion whose composition is AlSi3O10 is 3 b The structure o...View the full answer

Answered By

SHINKI JALHOTRA
I have worked with other sites like Course Hero as a tutor and I have great knowledge on IT skills.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 978-0134414232
14th Edition
Authors: Theodore Brown, H. LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward, Matthew Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
Determine the charge of the aluminosilicate ion whose composition is AlSi 3 O 10 .
-
Using Table 1.3, determine what charge the ion will carry when each of the following elements reacts to form an ionic compound: Al, Li, S, and O. Table 1.3 Table 1.3 Valence Electrons of the First 18...
-
Propose a reasonable structure for each of the following molecules on the basis of the given molecular formula and of the 1H and proton-decoupled l3C NMR data (a) C7H16O, spectra J and K (* = CH2 by...
-
Is the State of Michigan justified in advertising the prize amount as $1.586 billion? Explain.
-
Jurisdiction E spends approximately $7 million each winter on snow removal. The jurisdiction is considering adding a new income tax provision that would allow people to deduct the cost of snow...
-
People who gravitate toward marketing where they can use their strength in qualitative reasoning are shown in this Advise to also have blind spots where they are called upon to substantiate ideas...
-
Ormet Primary Aluminum Corporation, operated an aluminum smelter plant in Hannibal, Ohio. The facility ceased production in October 2013 in order to liquidate its assets after filing for bankruptcy...
-
Anna Broderick is the dietitian for the State University football team, and she is attempting to determine a nutritious lunch menu for the team. She has set the following nutritional guidelines for...
-
Create three new users with names and privileges as described below in Oracle database: Show the code you used to make these users and grant them the correct privileges. Name User1 User2 User3...
-
A ride hailing company has their DB structured in 3 major tables as described in the SCHEMA section below. Write a query to fetch the top 100 users who traveled the most distance using the service....
-
An Iron corrodes to produce rust, Fe 2 O 3 , but other corrosion products that can form are Fe(O)(OH), iron oxyhydroxide, and magnetite, Fe 3 O 4 . (a) What is the oxidation number of Fe in iron...
-
Which of the following statements are true? (a) Si can form an ion with six fluorine atoms, SiF 6 2- , whereas carbon cannot. (b) Si can form three stable compounds containing two Si atoms each, Si 2...
-
High blood pressure results from constriction of the arteries. To maintain a normal flow rate (flux), the heart has to pump harder, thus increasing the blood pressure. Use Poiseuilles Law to show...
-
Respond to your classmate. . If you have software or something running on the operating system, those logs can be found in other locations. These logs are generally stored in plain text and things...
-
How would you put this in Statement of Cash Flows Reconciliation to Net Income, Statement of Financial Position, and Statement of Operations? 1/1/2022 Debit Credit Cash $ 950,000.00 Common Stock $...
-
Replace the force system with a single resultant force. Let F = [F] KN. e) Determine the point y on the y-axis through which the resultant must pass. 8 kN-m 3 m 5 m F 13/12 4 m A 4 m P 3 m 4 kN 60
-
A capacitor has parallel plates that have an area of 1.06 cm and are 1.04 mm apart. There is a vacuum between the plates, and the electric field magnitude is 1.09 x 106 N/C. (a) Find the potential...
-
The following data are from Kelloggs 10-K report dated December 29, 2018 ($ millions). Revenue $13,547 Earnings from continuing operations $1,344 Interest expense 287 Capital expenditures (CAPEX) 578...
-
Suppose the production of airframes is characterized by a CES production function: Q = (L + K)2. The marginal products for this production function are MPL = (L + K)L and MPK = (L+ K)K. Suppose that...
-
Bonus shares can be issued out of revenue reserves. True/False?
-
(a) Based on the lattice energies of MgCl2 and SrCl2 given in Table 8.2, what is the range of values that you would expect for the lattice energy of CaCl2? (b) Using data from Appendix C, Figure 7.9,...
-
(a) What is meant by the term covalent bond? (b) Give three examples of covalent bonding. (c) A substance XY, formed from two different elements, boils at -33oC. Is XY likely to be a covalent or an...
-
Which of these elements are unlikely to form covalent bonds: S, H, K, Ar, Si? Explain your choices.
-
1. Country A and B each have 2000 units of labour. In Country A, one unit of labour can produce 7 kilograms of cheese or 42 kilograms of coffee. In Country B, one unit of labour can produce 14...
-
Suppose Andrew is deciding on buying a new bike for exercise. There are three periods: Period 0 Andrew decides whether to buy the bike or not. The cost of the bike is equal to P utility units....
-
To be a case manager provide a detailed short summary including your intentions and goals and the type of opportunities that are helpful for securing a position as a case manager. Write it in the...

Study smarter with the SolutionInn App


