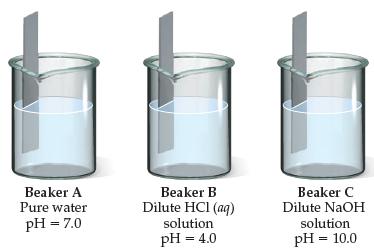
Bars of iron are put into each of the three beakers as shown here. In which beakerA,
Question:
Bars of iron are put into each of the three beakers as shown here. In which beaker—A, B, or C—would you expect the iron to show the most corrosion?
Transcribed Image Text:
Beaker A Pure water Beaker B Dilute HCI (aq) solution Beaker C Dilute NaOH pH = 7.0 solution pH = 4.0 pH = 10.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
The Evolution of the Fashion Industry: Consider the market for clothes and suppose there exist 100 different styles that can be produced and can be arranged (and equally spaced) on a circle. Among...
-
In the U.S., Presidential elections are not won by the candidate who wins the popular vote nationally. (If they were won in this way, Al Gore would have become President in 2000.) Rather, each state...
-
In cases 1-3 below, assume that Division A has a product that can be sold either to Division B of the same company or to outside customers. The managers of both divisions are evaluated based on their...
-
What is GST?
-
What is meant by indirect least squares? When is it used?
-
Design the following filters using the Kaiser window: (a) \(A_{\mathrm{p}}=1.0 \mathrm{~dB}\) \(A_{\mathrm{r}}=40 \mathrm{~dB}\) \(\Omega_{\mathrm{p}}=1000 \mathrm{rad} / \mathrm{s}\)...
-
List, describe, and provide examples of the various defenses available to a defendant charged with a business crime.
-
The following selected accounts and their current balances appear in the ledger of Clairemont Co. for the fiscal year ended May 31, 2019: Selected accounts and related amounts for Clairemont Co. for...
-
When an object is placed between the pole(P) and focus(F) of a concave mirror, the image formed is (i) behind the mirror (ii) virtual and erect and (iii) larger than the object (or magnified) M A' E...
-
Uric acid is degraded by uricase enzyme immobilized in porous Ca-alginate beads. Experiments conducted with different bead sizes result in the following rate data: a. Determine the effectiveness...
-
The electrodes in a silver oxide battery are silver oxide (Ag 2 O) and zinc. Which electrode acts as the anode?
-
In each of the following balanced oxidationreduction equations, identify those elements that undergo changes in oxidation number and indicate the magnitude of the change in each case. 2MnO 4 - (aq) +...
-
College students trying to register for a course sometimes find that the course has been closed, or the section they want has been closed. Prepare a cause-and-effect diagram for this problem.
-
Anke Perks is opening an arts and crafts store that focuses on canvases, paints, and pencils. Each canvas is sold for $24. The variable operating costs are $14 per canvas while the fixed operating...
-
What are the political and economic factors that have led to the emergence of social entrepreneurship as a promising method of addressing social ills?
-
Why do social ventures pursuing both economic and social purposes often experience identity tension?
-
A three-phase \(25-\mathrm{kVA}, 480-\mathrm{V}, 60-\mathrm{Hz}\) alternator, operating under balanced steady-state conditions, supplies a line current of \(20 \mathrm{~A}\) per phase at a 0.8...
-
A stranded conductor is an example of a composite conductor. (a) True (b) False
-
Refer to Practice 20-5. Prepare the comparative note disclosure that would be provided in the notes to the 2013 financial statements with respect to the income statements for 2011, 2012, and 2013.
-
What is a manufacturing system?
-
Chlorine is widely used to purify municipal water supplies and to treat swimming pool waters. Suppose that the volume of a particular sample of Cl2 gas is 8.70 L at 895 torr and 24oC. (a) How many...
-
Many gases are shipped in high-pressure containers. Consider a steel tank whose volume is 55.0 gallons that contains O2 gas at a pressure of 16,500 kPa at 23oC. (a) What mass of O2 does the tank...
-
In an experiment reported in the scientific literature, male cockroaches were made to run at different speeds on a miniature treadmill while their oxygen consumption was measured. In one hour the...
-
3. The Alice Company has estimated costs for the following monthly production levels: Production volume (units) Cost A Cost B Cost C Cost D Cost E Cost F Cost G Cost H Total manufacturing costs...
-
ted eted eted eted (20%) Problem 3: Consider a circuit shown in the figure. Ignore the internal resistances of the batteries. R3 Randomized Variables A LL F E = 46 V 82=48 V R = 8Q R = 8Q R B E...
-
A 40.0-g object connected to a spring with a force constant of 50.0 N/m oscillates with an amplitude of 6.00 cm on a frictionless, horizontal surface. (a) Find the total energy of the system. mJ (b)...

Study smarter with the SolutionInn App


