Imagine the primitive cubic lattice. Now imagine grabbing opposite corners and stretching it along the body diagonal
Question:
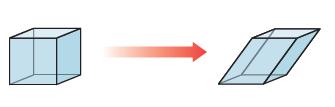
Imagine the primitive cubic lattice. Now imagine grabbing opposite corners and stretching it along the body diagonal while keeping the edge lengths equal. The three angles between the lattice vectors remain equal but are no longer 90°. What kind of primitive lattice have you made?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
When you stretch a cubic lattice along its body diagonal while keeping the edge lengths equal you ...View the full answer

Answered By

Isabel Seraspi
I have experience teaching math, science, and English to students of all ages. I have also worked as a tutor in a college setting, helping students with their homework and preparing them for exams.
I believe that tutoring is a great way to help students learn. It allows students to get one-on-one help with their studies, and it gives them the chance to ask questions and get immediate feedback. Tutoring can also be tailored to the individual needs of the student, which is why I believe it is so effective.
I have seen firsthand how tutoring can help students improve their grades and confidence. I have also seen how it can help students who are struggling with a particular subject. I believe that tutoring is a great way to help students learn and succeed in school.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 978-0134414232
14th Edition
Authors: Theodore Brown, H. LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward, Matthew Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
Imagine the primitive cubic lattice. Now imagine grabbing the top of it and stretching it straight up. All angles remain 90. What kind of primitive lattice have you made?
-
Imagine the primitive cubic lattice. Now imagine pushing on top of it, straight down. All angles remain 90. What kind of primitive lattice have you made?
-
Three objects of equal mass are located at three corners of a square of edge length l as in Figure P13.23. Find the gravitational field at the fourth corner due to these objects. M2 I2L Figure P13.23
-
The figure below represents a schematic of pipe network. A rate of 35 Ls, is pumped to feed two lines (3-4-5-6; and 2-7-8). The length and diameter of each pipe segment are listed in the table....
-
Curtis Bedford, age 73, is a professor of English at a private university. He holds a tenured position, which represents a lifetime employment contract. The university has offered Curtis a $75,000...
-
Use the figure to work Problem shows the marginal social benefit and marginal social cost of a waste disposal system in a city of 1 million people. If voters are well informed about the costs and...
-
In 1951, DuPont began using the chemical perfluorooctanoic acid to manufacture Teflon. Due to the dangerous nature of the chemical, DuPont was given special instructions by its supplier to dispose of...
-
Presented below is information related to equipment owned by Suarez Company at December 31, 2007. Cost ............... $9,000,000 Accumulated depreciation to date ... 1,000,000 Expected future net...
-
8. Determine one possible value for x when 0x2 for each of the following. Give answers in simplest exact form. [A6] a. |sinh = sinx, x #23 3/4 b. cos _1131* =sinx c. tan =cotx I
-
The following is the pre-closing trial balance for Christina Rehabilitation Hospital as of September 30, 2020. Required a. Prepare a statement of operations and a statement of changes in net assets...
-
Indicate the type of solid (molecular, metallic, ionic, or covalent-network) for each compound: (a) CaSO 4 , (b) Pd, (c) Ta 2 O 5 (melting point, 1872 C), (d) Caffeine (C 8 H 10 N 4 O 2 ), (e)...
-
Covalent bonding occurs in both molecular and covalent-network solids. Which of the following statements best explains why these two kinds of solids differ so greatly in their hardness and melting...
-
Find 95% confidence intervals in the following cases, assuming that the standard deviations are known to match those in the earlier problem. Does the confidence interval include the true mean? 1. A...
-
if you want to triple your money at an interest rate of 8 percent per year compounded annually, how many years would you have to leave the money in the account?
-
On December 1, 2016 fine dining products borrowed $88000 on a 11%, 8 note with annual installment payments of $11000 plus interest due on December 1 of each succeeding year. On December 1 the...
-
company emerson purchased an office building for $2,530,000. the estimated useful life of the building is 40 years. what is the monthly amount of depreciation expense computed by the straight line...
-
Which of the options below best describes the properties of polymeric materials? a. The presence of crosslinks between the chains is responsible for the high ductility of thermosets b. Thermosets...
-
In a lottery game where 3 numbers are chosen from the set {0, 1, 2, 3, 4, 5, 6, 7, 8, 9}, how many different 3-number cards could be made where a) order is taken into consideration? b) order is not...
-
In Figure 11.8, the X and Y coordinates (in feet) of A are 4371.56 and 8987.63, those of B are 8531.05 and 8312.57, and those of C are 10,240.98 and 8645.07, respectively. Also angle x is 50o 12'45''...
-
Smiths Family Fashions implemented a balanced scorecard performance measurement system several years ago. Smiths is a locally owned clothing retailer with fashions for men, women, teens, and...
-
Consider the following reagents: zinc, copper, mercury (density 13.6 g/mL), silver nitrate solution, nitric acid solution. (a) Given a 500-mL Erlenmeyer flask and a balloon can you combine two or...
-
Lanthanum metal forms cations with a charge of 3+. Consider the following observations about the chemistry of lanthanum: When lanthanum metal is exposed to air, a white solid (compound A) is formed...
-
A 35.0-mL sample of 1.00 M KBr and a 60.0-mL sample of 0.600 M KBr are mixed. The solution is then heated to evaporate water until the total volume is 50.0 mL. What is the molarity of the KBr in the...
-
Let XI, X2 , X3 be independent N(I', 0' 2 ) variates. (a) Show that for IVI :S: 11'/6 we may write X( 3) = X( 2) + S cos V +v'3Ssin V, X( 1 ) = X( 2) - S cos V + v'3Ssin V, where (b) Show that so...
-
Your friend, Juan, is planning to buy a new car, which costs $21,399. Juan has a job that pays pretty well and a good credit record, but hes never bought a car before, and he doesnt want to get...
-
Calculating the promatic and enthumi wetge note of return the common stock of the Banga Cutie Company had the obowing and paar hock perloes ver the last fire pour and paidro cash andere Bags 316 10 3...

Study smarter with the SolutionInn App


