The free-base form of cocaine (C 17 H 21 NO 4 ) and its protonated hydrochloride form
Question:
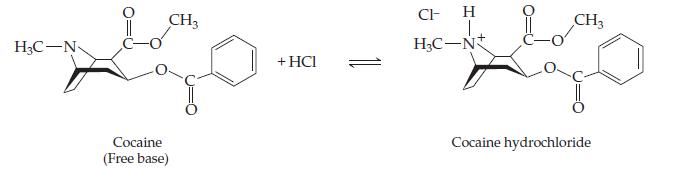
The “free-base” form of cocaine (C17H21NO4) and its protonated hydrochloride form (C17H22ClNO4) are shown below; the free-base form can be converted to the hydrochloride form with one equivalent of HCl. For clarity, not all the carbon and hydrogen atoms are shown; each vertex represents a carbon atom with the appropriate number of hydrogen atoms so that each carbon makes four bonds to other atoms.
(a) One of these forms of cocaine is relatively water-soluble: which form, the free base or the hydrochloride?
(b) One of these forms of cocaine is relatively insoluble in water: which form, the free base or the hydrochloride?
(c) The free-base form of cocaine has a solubility of 1.00 g in 6.70 mL ethanol (CH3CH2OH). Calculate the molarity of a saturated solution of the free-base form of cocaine in ethanol.
(d) The hydrochloride form of cocaine has a solubility of 1.00 g in 0.400 mL water. Calculate the molarity of a saturated solution of the hydrochloride form of cocaine in water.
(e) How many mL of a concentrated 18.0 M HCl aqueous solution would it take to convert 1.00 kilograms (a “kilo”) of the free-base form of cocaine into its hydrochloride form?
Step by Step Answer:

Chemistry The Central Science
ISBN: 978-0134414232
14th Edition
Authors: Theodore Brown, H. LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward, Matthew Stoltzfus





