The orbital diagram that follows presents the final step in the formation of hybrid orbitals by a
Question:
The orbital diagram that follows presents the final step in the formation of hybrid orbitals by a silicon atom. What type of hybrid orbital is produced in this hybridization?
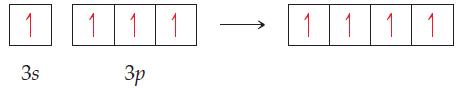
Transcribed Image Text:
1 111 1111 3s Зр
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
On excitation an electron from 3sorbital prom...View the full answer

Answered By

Umair Expert
Hi Everyone.
I have 6 years of teaching experience.
I am serving as a tutor 2 more websites.
I will provide you projects and solutions of questions related to any subject.
I am a good programmer.(PHP, Python)
I am good command in mathematics.
I am here to assist you.
I'll be very happy to work with you...
Thanks dear..
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Question Posted:
Students also viewed these Sciences questions
-
(a) Starting with the orbital diagram of a sulfur atom, describe the steps needed to construct hybrid orbitals appropriate to describe the bonding in SF2. (b)What is the name given to the hybrid...
-
The orbital diagram that follows shows the valence electrons for a 2+ ion of an element. (a) What is the element? (b) What is the electron configuration of an atom of this element? 4d
-
(a) Starting with the orbital diagram of a boron atom, describe the steps needed to construct hybrid orbitals appropriate to describe the bonding in BF3. (b)What is the name given to the hybrid...
-
is manager should spend time in understanding the market before taking decisions? If yes, why?
-
The following is a stem-and-leaf display with two-digit leaves. (The leading leaf digit = 10.0.) List the corresponding measurements. 0 313 974 8468 6426 4954 13 41001 123456
-
Mary was interested in comparing the mean word length in articles from a medical journal and an airlines in-flight magazine. She counted the number of letters in the first 400 words of an article in...
-
Consider the delivery time data discussed in Example 11.3. Find the PRESS statistic for the model developed from the estimation data. How well is the model likely to perform as a predictor? Compare...
-
Was the confiscation of all cigar manufacturers by the Cuban government in the early 1960s and expropriation or a nationalization? Was this confiscation commercial activity in which a private...
-
1. Fill in the following information below: Assets Liabilities Stockholder's Equity Business #1 ? $181,000 $212,000 Business #2 $74,800 ? $36,200 Business # 3 $30,000 $21,400 ? 2. Each of the...
-
Parent Company owns 60 percent of the voting common stock of the Subsidiary Inc. On January 1, 2019, the Subsidiary sold ten-year bonds with a face value of $560,000 to the public for $604,000. The...
-
(a) If these three balloons are all the same size, what angle is formed between the red one and the green one? (b) If additional air is added to the blue balloon so that it gets larger, what happens...
-
What property of the electron causes electron domains to have an effect on molecular shapes?
-
(a) Show that the spectral density of the radiant energy flux the arrives in the solid angle d is cu cos d/4, where is the angle the normal to the unit area makes with the incident ray, and u is...
-
Reimplement the LinkedList class of Section 16.1.8 so that it implements the java.util.List interface. Extend the java.util.AbstractSequentialList class.
-
Describe a recursive algorithm for finding the total path length of a tree. The total path length is the sum of the lengths of all paths from the root to the leaves. (The length of a path is the...
-
Suppose you are stranded on a desert island on which stacks are plentiful, but you need a queue. How can you implement a queue using two stacks? What is the big-Oh running time of the queue...
-
Why does Figure 21 show all possible configurations of a double-red violation? Figure 21 13 722 13 5 t 13. t 12 t
-
Write a method: public static void reverse that reverses the entries in a linked list. (LinkedList strings)
-
Amber's employer, Lavender, Inc., has a 401(k) plan that permits salary deferral elections by its employees. Amber's salary is $99,000, and her marginal tax rate is 33%. a. What is the maximum...
-
The first national bank pays a 4% interest rate compound continuously. The effective annual rate paid by the bank is __________. a. 4.16% b. 4.20% c. 4.08% d. 4.12%
-
Repeat Prob. 1756 for supersonic flow at the inlet.
-
Air enters a nozzle at 0.2 MPa, 350 K, and a velocity of 150 m/s. Assuming isentropic flow, determine the pressure and temperature of air at a location where the air velocity equals the speed of...
-
Repeat Prob. 1758 assuming the entrance velocity is negligible.
-
Create a class named Account that contains: A private int data field named id for the account (default 0). A private double data field named balance for the account (default 0). A private double data...
-
The next Java code needs to be converted from static to dynamic (just addd changes in same code): import java.util.Scanner; class MyIntStaticCircularQueue { int capacity = 2; int queue[] = new...
-
Hi! Would a tutor be able to assist me on this? In a complete graph with 48 vertices, how many vertices will be in each node's adjacency list? How many entries will be in the adjacency matrix for...

Study smarter with the SolutionInn App


