This figure shows the interaction of a cation with surrounding water molecules. Would you expect the energy
Question:
This figure shows the interaction of a cation with surrounding water molecules.
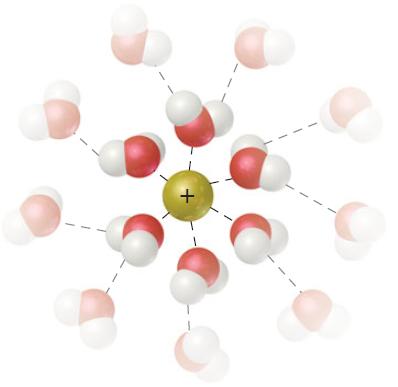
Would you expect the energy of ion–solvent interaction to be greater for Na+ or Li+? Explain.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
Ionsolvent interactions inversely proportion al to the size of the ion Ie smaller is ...View the full answer

Answered By

Umair Expert
Hi Everyone.
I have 6 years of teaching experience.
I am serving as a tutor 2 more websites.
I will provide you projects and solutions of questions related to any subject.
I am a good programmer.(PHP, Python)
I am good command in mathematics.
I am here to assist you.
I'll be very happy to work with you...
Thanks dear..
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Question Posted:
Students also viewed these Sciences questions
-
Which of the following molecules would you expect to be aromatic? (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) N+
-
Would you expect alanine (an amino acid) to be more soluble in water or in hexane? Explain.
-
The accompanying graph depicts the interaction energy between two water molecules situated so that their dipole moments are parallel and pointing in the same direction. Sketch an approximate curve...
-
The Federal Reserve may raise its benchmark interest rate later this month. How is this achieved? Why would they do this? Explain the consequences fully. (Include graphs with your answer)
-
Find and s for: (a) The lizard data in Exercise 2.19. (b) The acid rain data in Exercise 2.115. 1.28 1.36 1.24 2.47 1.94 2.52 2.67 1.29 1.56 2.66 2.17 57 2.10 2.54 163 2.11 2.57 1.72 0.76 1.02 1.78...
-
Why does confirmation bias lead to defective decision making/problem solving?
-
In Example 8.6, suppose both people pull on the same end of the rope, each exerting a force \(F\), while the other end is still tied to the tree. Is the tension in the rope larger than, equal to, or...
-
Grant, Arthur, and David formed a partnership for the purpose of betting on boxing matches. Grant and Arthur would become friendly with various boxers and offer them bribes to lose certain bouts....
-
The correct syntax of the re . sub ( ) function is re . sub ( string , replacement, pattern ) re . sub ( pattern , string, replacement ) re . sub ( replacement , pattern, string ) re . sub ( pattern...
-
Nancy Tercek started a delivery service, Tercek Deliveries, on June 1, 2015. The following transactions occurred during the month of June. June 1 Stockholders invested $10,000 cash in the business in...
-
What molecular structural features cause high-density polyethylene to be denser than low-density polyethylene?
-
List four properties of a solution that depend on the total concentration but not the type of particle or particles present as solute.Write the mathematical expression that describes how each of...
-
Give three examples of industries that probably use operation costing.
-
b) Assume you are writing a test case for a Web Service. Consider following. Same Web Service needs to run for 1000 test data sets. Required commands are unique across all 1000 test data sets. Using...
-
Limited or constrained resources are a fact in virtually every project. Often there are one or two resources that may be especially difficult to manage, so a project manager needs to look carefully...
-
a house sale is being closed on august 1 . the fair market value of the property is 1 4 4 . 0 0 0 $ . the assessed value is 5 0 % of fair market value and the tax levy is 5 5 mills . the tax paid in...
-
Institute training on the job. Questions: 1. What kinds of training would be useful? 2. Has training ever been given in the past that proved useless? Was the training itself at fault, or did the boss...
-
Of the 9 elements suggested by the SBA to be contained in a business plan, how many are solely focused on financial factors?
-
Two particles, A and B, have the same mass, but particle A has a charge of +q and B has a charge of +2q. The particles are accelerated from rest through the same potential difference. Which one has...
-
Why do bars offer free peanuts?
-
Air in an automobile tire is maintained at a pressure of 220 kPa (gauge) in an environment where the atmospheric pressure is 94 kPa. The air in the tire is at the ambient temperature of 25C. Now a...
-
The thrust developed by the engine of a Boeing 777 is about 380 kN. Assuming choked flow in the nozzle determine the mass flow rate of air through the nozzle. Take the ambient conditions to be 265 K...
-
A stationary temperature probe inserted into a duct where air is flowing at 250 m/s reads 85C. What is the actual temperature of air?
-
Regarding the following structure answer the following questions: N 1) What is the sar? wwww olaparib (lynparza) 2)What is the quantitive estimation (principle of assay)?
-
7. Determine the Q-values of the following reaction: N+13N P+13C (Given: Rest-mass of 13N = 13.005739 u; rest-mass of 13C = 13.003355 u.) (3 marks)
-
Consider the following description for the element iron, Fe Ph Iron is a silver-white malleable ductile magnetic heavy metallic element that readily rusts in moist air to give a hydrated iron oxide....

Study smarter with the SolutionInn App


