Which arrangement of atoms in a lattice represents close-packing? (i)
Question:
Which arrangement of atoms in a lattice represents close-packing?
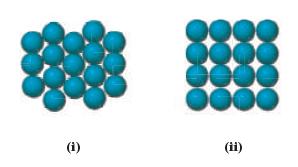
Transcribed Image Text:
(i)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
ii In a closepacked lattice the atoms are arranged in such a way as to maximize the packing effi...View the full answer

Answered By

Isabel Seraspi
I have experience teaching math, science, and English to students of all ages. I have also worked as a tutor in a college setting, helping students with their homework and preparing them for exams.
I believe that tutoring is a great way to help students learn. It allows students to get one-on-one help with their studies, and it gives them the chance to ask questions and get immediate feedback. Tutoring can also be tailored to the individual needs of the student, which is why I believe it is so effective.
I have seen firsthand how tutoring can help students improve their grades and confidence. I have also seen how it can help students who are struggling with a particular subject. I believe that tutoring is a great way to help students learn and succeed in school.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 978-0134414232
14th Edition
Authors: Theodore Brown, H. LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward, Matthew Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
How does the arrangement of atoms in a crystalline substance differ from the arrangement in a noncrystalline substance?
-
Atoms are found to move from one lattice position to another at the rate of 5 105 jumps/s at 400 C when the activation energy for their movement is 30,000 cal/mol. Calculate the jump rate at 750 C?
-
How does the number of atoms in a 26.5-gram gold ring compare to the number in a silver ring of the same mass?
-
Which one the below does not define "Work role boundaries" of a care worker limits that allow a patient and staff to connect safely in a therapeutic relationship based on patients' needs rules of...
-
Mrs. F, age 57, participates in the group-term life insurance plan sponsored by her corporate employer. According to Treasury tables, the cost of $1,000 of life insurance for a 57-year-old person is...
-
PepsiCos financial statements are presented in Appendix B. Financial statements for Thc Coca-Cola Company are presented in Appendix C. Instructions for accessing and using the complete annual reports...
-
What stakeholder affected by their behavior was Gilead weighting very lightly when it decided what to tell the FDA about the medicines it wished to have permission to sell?
-
Recognition of Profit, Percentage-of-Completion In 2010 Gurney Construction Company agreed to construct an apartment building at a price of $1,200,000. The information relating to the costs and...
-
The BiHye Cycle Company has three plants that supply deluxe bicycles to three main countries of USA, Canada, and Germany. The plants' daily capacities, the countries' yearly requirements, as well as...
-
A bakery makes three types of cookies, I, II, and III. Each type of cookie is made from the four ingredients A, B, C, and D. The number of units of each ingredient used in each type of cookie is...
-
The unit cell of a compound containing potassium, aluminum, and fluorine is shown here. (a) What type of lattice does this crystal possess (all three lattice vectors are mutually perpendicular)? (b)...
-
Indicate whether each statement is true or false: (a) The liquid crystal state is another phase of matter, just like solid, liquid, and gas. (b) Liquid crystalline molecules are generally spherical...
-
Each of the following situations has an internal control weakness. a. Betty Grable has been your trusted employee for 30 years. She performs all cash-handling and accounting duties. Betty Grable just...
-
Describe the mechanism and regulation of ketogenesis. When is ketogenesis the highest? Is ketosis safe long-term?
-
Which method of allocating service department costs (the direct method, the step method, and the reciprocal method) would result in you potentially receiving a higher year-end bonus?
-
Albatross Services scrapped a van. The van originally cost $40,000, had an estimated salvage value of zero, and an estimated life of 10 years. At the time it was scrapped, it had accumulated...
-
Sams building that was used for business was partially destroyed by a hurricane. There was no insurance for hurricane damage. The building had an adjusted basis of $46,000. The FMV was $90,000 just...
-
The concept of "Power Payments" will help you repay your loans faster and reduce your total interest payments. Consider a situation where you have 3 loans: Loan A has a loan payment of $500/month and...
-
For the subdivision in the accompanying figure, assume that lines AC, DF, GI, and JL are parallel, but that lines BK and CL are parallel to each other, but not parallel to AJ. If the X and Y...
-
Cleaning Service Company's Trial Balance on December 31, 2020 is as follows: Account name Debit Credit Cash 700 Supplies Pre-paid insurance Pre-paid office rent Equipment Accumulated depreciation -...
-
(a) Calculate the standard enthalpy of formation of gaseous diborane (B2H6) using the following thermochemical information: (b) Pentaborane (B5H9) is another boron hydride. What experiment or...
-
From the following data for three prospective fuels, calculate which could provide the most energy per unit volume: Density at 20 C Molar Enthalpy of Combustion Fuel (g/cm (kJ/mol) Nitroethane, C2H...
-
The hydrocarbons acetylene (C2H2) and benzene (C6H6) have the same empirical formula. Benzene is an "aromatic" hydrocarbon, one that is unusually stable because of its structure. (a) By using the...
-
what ways do visionary leaders cultivate a culture of creativity and foresight within their organizations?
-
The decisions to establish a deferred tax asset valuation allowance and, if so, what amount to record are subjective assessments. Readily observable criteria do not exist, and the dollar amounts can...
-
How do visionaries anticipate and navigate emergent trends and paradigm shifts in complex socio-economic landscapes ?

Study smarter with the SolutionInn App


