The chart above shows the main types of radiation in the electromagnetic spectrum. Two haven't been named.
Question:
The chart above shows the main types of radiation in the electromagnetic spectrum. Two haven't been named. Which type does P represent?
A. Microwaves
B. Sound waves
C. Gamma rays
D. Ultraviolet
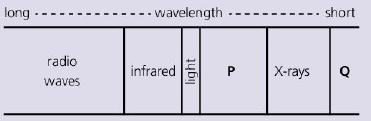
Transcribed Image Text:
long - -wavelength -- - short radio infrared X-rays waves light P.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
D Ultraviolet From ...View the full answer

Answered By

Anzila Parveen c p
Am anzila 25 y/o lady physics graduated and interested to solve your questions mainly about physics . I teach many students at my home . And make physics very simple to them . I hope i can share my knowledge with you.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following diagrams represent two electromagnetic waves. Which wave corresponds to the higher-energy radiation? Explain. a. b.
-
GAMMA produces over a hundred different types of residential water faucets at its Delta, Florida, plant. This plant uses activity- based costing to calculate product costs. The following table...
-
The wavelength of absorbed electromagnetic radiation for CoBr42- is 3.4 X 10-6 m. Will the complex ion CoBr64- absorb electromagnetic radiation having a wavelength longer or shorter than 3.4 X 106 m?...
-
Find a vector parametrization of the intersection of the surfaces x 2 + y 4 + 2z 3 = 6 and x = y 2 in R 3 .
-
Draw resonance formulas of the nitric acid molecule, HNO3. What is the geometry about the N atom? What is the hybridization on N? Use bond energies and one Lewis formula for HNO3 to estimate Hf for...
-
How long would it take a 4.50-kW motor to raise a 175-kg boiler to a platform 15.0 m above the floor?
-
John J. Williams began working at Delaware Elevator as a branch sales manager in late 2004. The company manufactures components for elevators and installs and repairs elevators. It employs 175 people...
-
The following information comes from the accounts of Jersey Company. Required a. There were $170,000 in sales on account during the accounting period. Write-offs of uncollectible accounts were...
-
Total Energy (a) Conserved 12. Which one of the following is true in the case of inelastic collisions? Kinetic Energy Conserved Momentum Conserved (b) Conserved Not Conserved Conserved (c) Conserved...
-
5.18 Length of a movie on Netflix. Flixable reports that Netflix's U.S. catalog contains almost 4000 movies.2 You are interested in determining the average length of these movies. Previous studies...
-
The diagram shows an aeroplane of mass 3.4 x 10 5 kg accelerating uniformly from rest along a runway. After 26 s it reaches a speed of 65 m/s. a. Calculate i. The acceleration of the aeroplane ii....
-
The diagram below shows a driver 50 m below the surface of the water. a. The density of water is 1000 kg/m 3 and the acceleration of free fall is 10 m/s 2 . Calculate the pressure that the water...
-
Many medical PET scans use the isotope \({ }^{18} \mathrm{~F}\), which has a half-life of \(1.8 \mathrm{~h}\). A sample prepared at 10:00 A.M. has an activity of \(20 \mathrm{mCi}\). What is the...
-
Imagine that your company is considering the launch of a product or brand in a new market. What company and product are you choosing? SWOT analysis for the company. Strengths In light of the...
-
For the distributed system shown in the figure below. a0 b0 CO al bl cl a2 a3 b2 62 2 20 33 b3 c3 P P time (1) Provide all the pairs of events that are related. (2) Provide logical time for all the...
-
In order to remain solvent and continue to support Americans, what are the goals of the Social Security Administration? Group of answer choices to delay the payouts to Americans by making them wait...
-
In the current year, Drake, a disabled taxpayer, made the following home improvements: Cost Pool installation, which qualified as a medical expense and increased the value of the home by $25,000....
-
How do you know if you would be a good IT project manager? After all, being an IT project manager is a demanding yet rewarding endeavor. Imagine you are chosen to be the IT project manager and must...
-
Find an upper triangular matrix that satisfies 3 1 30
-
Choose two matrices A and B with dimension 2 x 2. Calculate det A, det B, and det (AB). Repeat this process until you are able to discover how these three determinants are related. Summarize your...
-
When 1-methylcyclohexene is treated with HCl, a Markovnikov addition is observed. How would you use 1 H NMR spectroscopy to determine that the major product is indeed the Markovnikov product?
-
How many signals will be expected in the 1 H NMR spectrum of each of the following compounds? (a) (b) (c) (d) (e) (f) (g) (h) `NO2
-
Compare the structures of ethylene, acetylene, and benzene. Each of these compounds produces only one signal in its 1 H NMR spectrum. Arrange these signals in order of increasing chemical shift. -...
-
Assume the following for your corporation: sales (aka revenue) = $403 Cost of goods sold = 160 depreciation = 35 Interest Expense = 20 tax rate = 40% What is the corporation's total after tax net...
-
Megan Furlan uses an automobile in a business that she carries on as a sole proprietor that uses a December 31 fiscal period. The automobile cost $88,000 in 2022, with maximum CCA being claimed for...
-
What type of Operating System would you use if you were asked to develop a system that processed 250 million credit card transactions each night? Why? 2. List two metrics, explain what they measure,...

Study smarter with the SolutionInn App


