Rank the following molecules from least oxidized to most oxidized: Ethane Ethanol ||
Question:
Rank the following molecules from least oxidized to most oxidized:
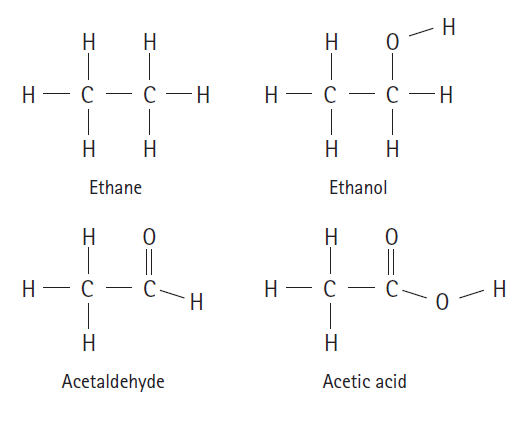
Transcribed Image Text:
Н Н Н Н — с — с —Н Н— с — с —Н Н Н Ethane Ethanol Н Н || Н — с- Н — с — С—н Н Н Н Acetaldehyde Acetic acid
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
From left to right these compounds are presented in order of least oxidized ...View the full answer

Answered By

Grace Igiamoh-Livingwater
I am a qualified statistics lecturer and researcher with an excellent interpersonal writing and communication skills. I have seven years tutoring and lecturing experience in statistics. I am an expert in the use of computer software tools and statistical packages like Microsoft Office Word, Advanced Excel, SQL, Power Point, SPSS, STATA and Epi-Info.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Conceptual Physical Science
ISBN: 978-0134060491
6th edition
Authors: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Physics questions
-
Rank the following molecules in order of increasing boiling point (without looking up the real values!): (a) 3-methylheptane; (b) Octane; (c) 2,4-dimethylhexane; (d) 2,2,4-trimethylpentane.
-
Rank the following molecules in order of the phase they form at room temperature: solid, liquid, gas. a. b. CH 3 CH 2 CH 2 CH 3 c. CH 3 CH 2 CH 2 CH 2 - OH CH H;C- C-
-
Rank the mass of these molecules from most to least. H. H. (. B A
-
Based on the information in Problems 9 and 10, what is Ryan and Nicoles liquidity ratio? What is their debt to asset ratio? Comment on each ratio. In Problems 10 Mortgage......... $ 43,500 Car...
-
Walter, who is single, owns a personal residence in the city. He also owns a cabin near a ski resort in the mountains. He uses the cabin as a vacation home. In August, Walter borrowed $60,000 on a...
-
Evaluate the given expressions using the property b logbx = x. 5(10 2 log 3 )
-
A hydroprocessing reactor, an asset used in petroleum refining, is placed into service at a cost of $2.7 million. It is thought to have a useful life, with turnarounds and proper maintenance, of 18...
-
SallyMay, Inc., designs and manufactures T-shirts. It sells its T-shirts to brand-name clothes retailers in lots of one dozen. SallyMays May 2013 static budget and actual results for direct inputs...
-
Problem 3. In class, we learned the Fisher Hypothesis: Treal From + E(i) It states that the real interest rate is the nominal interest rate minus the expected inflation. We observe inflation. We...
-
You are a senior staff member in the finance department of BCom Investments Ltd (BCom). In your role, you are charged with reviewing, performing analysis and making recommendations to the President...
-
An acid and a base react to form a salt, which consists of positive and negative ions. Which forms the positive ions: the acid or the base? Which forms the negative ions?
-
Review the concept of electronegativity in Section 15.6, and rank these elements from the weakest to strongest reducing agent: (a) chlorine, Cl; (b) sulfur, S; (c) sodium, Na.
-
Fordham Corporation produces a single product. The standard costs for one unit of its Concourse product are as follows: Direct materials (6 pounds at $0.50 per pound) . $ 3 Direct labor (2 hours at...
-
The facts of the case are simple: A dispute in the shop at an auto parts manufacturing plant in Hamilton, Iowa, ended when one worker murdered another. At 2:00 p.m. police responded to a report of a...
-
What are there penalties for companies like Starbucks that refuse employees the right to unionize? Explain briefly
-
Explain the communication process (communication loop) with diagram and examples
-
Your organization is a chemical supplier with employees in the following jurisdictions: Manitoba Prince Edward Island Yukon The organization is planning to implement a company-wide policy with...
-
How can strategic management within the HRM department ensure that HRM plays an effective role in the company's strategic management process? What are some of the key environmental variables that...
-
Determine whether each function's vertex is a maximum point or a minimum point and find the coordinates of this point. Find the zeros, if any exist, and the y-intercept. Then sketch the graph of the...
-
Sue Deliveau opened a software consulting firm that immediately paid $2,000 for a computer. Was this event a transaction for the business?
-
For the pump having the characteristics shown in Problem 14.14, operating at maximum efficiency with the speed increased to 1000 rpm, what will be (a) the new discharge flow rate and (b) the power...
-
The pump having the characteristics shown in Problem 14.14 is to be operated at 800 rpm. What discharge rate is to be expected if the head developed is 410 m? Data From Problem 14.14 Performance...
-
If the pump having the characteristics shown in Problem 14.14 is tripled in size but halved in rotational speed,what will be the discharge rate and head when operating at maximum efficiency? Data...
-
1. What is the RMA Industry's ROE? (Answer in terms of %, so 1.23% would just be 1.23). 2. For FYE 2022, what was Steelcase's ROA? (Answer in terms of %, so 1.23% would just be 1.23). 3. For FYE...
-
How does the poet's adept use of imagery serve to transmute the ineffable essence of human experience into a vivid tapestry of sensory perception and emotional resonance?
-
If daily demand is constant at 10 units per day, and lead time averages 12 days with a standard deviation of 3 days, 95 percent service requires how much safety stock?

Study smarter with the SolutionInn App


